All Photos(2)
About This Item
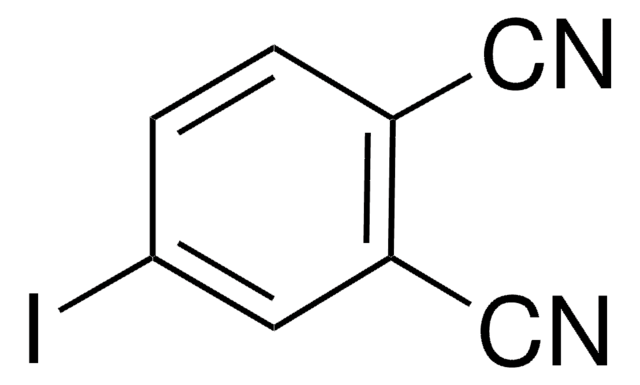
Linear Formula:
O2NC6H3(CN)2
CAS Number:
Molecular Weight:
173.13
Beilstein:
2263686
MDL number:
UNSPSC Code:
12171500
PubChem Substance ID:
NACRES:
NA.47
Recommended Products
Quality Level
Assay
99%
form
solid
mp
162-165 °C (lit.)
application(s)
diagnostic assay manufacturing
hematology
histology
storage temp.
room temp
SMILES string
[O-][N+](=O)c1cccc(C#N)c1C#N
InChI
1S/C8H3N3O2/c9-4-6-2-1-3-8(11(12)13)7(6)5-10/h1-3H
InChI key
UZJZIZFCQFZDHP-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Taner Arslan et al.
Journal of fluorescence, 30(2), 365-373 (2020-02-23)
In present work, a new substituted phthalonitrile derivative was prepared by the nucleophilic displacement reaction and then highly soluble zinc phthalocyanine (ZnPc) with four peripheral 1-hydroxyhexan-3-ylthio groups was synthesized by cyclotetramerization and characterized by FTIR, 1H and 13C NMRs spectroscopies
G S Amitha et al.
Journal of fluorescence, 29(1), 279-291 (2019-01-10)
A novel 4,4'-fluoresceinoxy bisphthalonitrile FPN is synthesized from fluorescein and 4-nitrophthalonitrile by aromatic nucleophilic ipso nitro substitution reaction. The structure of FPN constitutes phthalonitrile-fluorescein-phthalonitrile, acceptor-donor-acceptor, A-D-A form and the solvatochromic study of newly synthesized compound FPN was done in hexane
Armağan Günsel et al.
Dalton transactions (Cambridge, England : 2003), 48(39), 14839-14852 (2019-09-26)
This study describes the synthesis and characterization of a non-peripherally tetra-substituted copper(ii) phthalocyanine bearing 4-(trifluoromethoxy)phenol groups. Some spectroscopic techniques such as FT-IR, 1H-NMR, 13C-NMR, MALDI-TOF, SEM and UV-vis were used to characterize the compounds. The novel molecule of 3-(4-(trifluoromethoxy) phenoxy)
Min Wang et al.
Nature communications, 10(1), 3602-3602 (2019-08-11)
Molecular catalysts that combine high product selectivity and high current density for CO2 electrochemical reduction to CO or other chemical feedstocks are urgently needed. While earth-abundant metal-based molecular electrocatalysts with high selectivity for CO2 to CO conversion are known, they
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service

![1,8-Diazabicyclo[5.4.0]undec-7-ene 98%](/deepweb/assets/sigmaaldrich/product/structures/120/564/5b373e23-1624-489c-8efb-692de0f96ffb/640/5b373e23-1624-489c-8efb-692de0f96ffb.png)