SMB00377
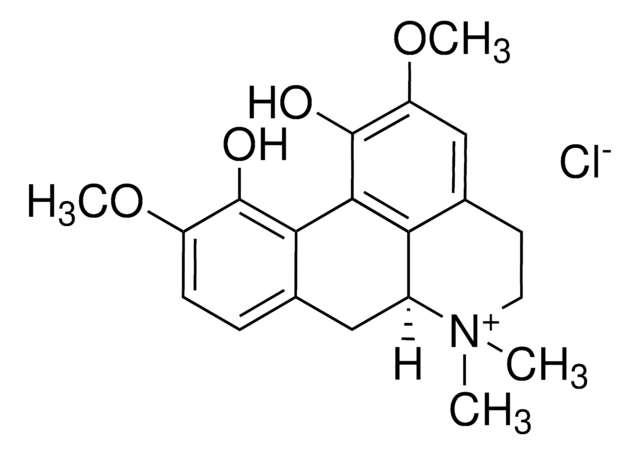
Magnoflorine
≥98% (HPLC)
Synonym(s):
(6aS)-2,10-dimethoxy-6,6-dimethyl-5,6,6a,7-tetrahydro-4H-dibenzo[de,g]quinoline-6-ium-1,11-diol
About This Item
Recommended Products
Quality Level
Assay
≥98% (HPLC)
form
powder
application(s)
metabolomics
vitamins, nutraceuticals, and natural products
storage temp.
2-8°C
SMILES string
OC1=C(OC)C=C2CC[N+](C)(C)[C@@]3([H])CC4=CC=C(OC)C(O)=C4C1=C32
InChI
1S/C20H23NO4/c1-21(2)8-7-12-10-15(25-4)20(23)18-16(12)13(21)9-11-5-6-14(24-3)19(22)17(11)18/h5-6,10,13H,7-9H2,1-4H3,(H-,22,23)/p+1/t13-/m0/s1
InChI key
YLRXAIKMLINXQY-ZDUSSCGKSA-O
Related Categories
General description
Application
Biochem/physiol Actions
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service