A1226
Anti-Actopaxin antibody produced in rabbit
IgG fraction of antiserum, PBS solution
Synonym(s):
Anti-α-Parvin, Anti-CH-ILKBP
About This Item
Recommended Products
biological source
rabbit
Quality Level
conjugate
unconjugated
antibody form
IgG fraction of antiserum
antibody product type
primary antibodies
clone
polyclonal
form
PBS solution
mol wt
antigen 42 kDa
species reactivity
human, mouse
technique(s)
microarray: suitable
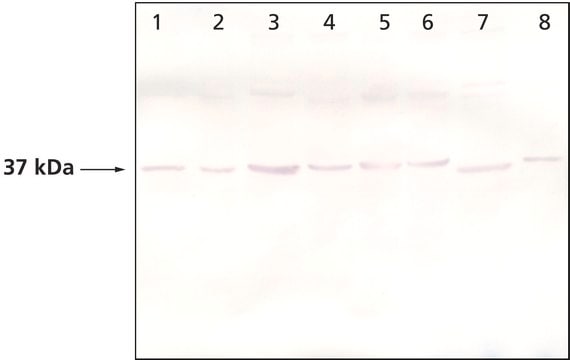
western blot: 1:3,000 using whole cell extract of the human endothelial ECV304 cell line.
UniProt accession no.
shipped in
dry ice
storage temp.
−20°C
target post-translational modification
unmodified
Gene Information
human ... PARVA(55742)
mouse ... Parva(57342)
General description
Rabbit anti-actopaxin recognizes actopaxin (42 kDa). Staining of actopaxin in immunoblotting is specifically inhibited with actopaxin immunizing peptide (mouse, amino acids 35-53).
Immunogen
Application
Immunofluorescence (1 paper)
Physical form
Disclaimer
Not finding the right product?
Try our Product Selector Tool.
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service