All Photos(2)
About This Item
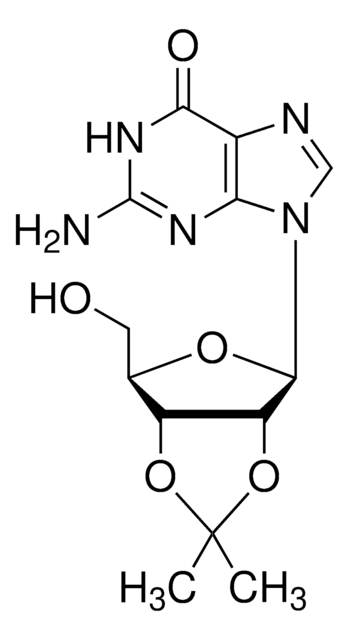
Empirical Formula (Hill Notation):
C13H17N5O4
CAS Number:
Molecular Weight:
307.31
Beilstein:
43435
EC Number:
MDL number:
UNSPSC Code:
12352204
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
optical activity
[α]20/D −98.5°, c = 1 in dioxane
mp
221-222 °C (lit.)
SMILES string
CC1(C)O[C@@H]2[C@@H](CO)O[C@H]([C@@H]2O1)n3cnc4c(N)ncnc34
InChI
1S/C13H17N5O4/c1-13(2)21-8-6(3-19)20-12(9(8)22-13)18-5-17-7-10(14)15-4-16-11(7)18/h4-6,8-9,12,19H,3H2,1-2H3,(H2,14,15,16)/t6-,8-,9-,12-/m1/s1
InChI key
LCCLUOXEZAHUNS-WOUKDFQISA-N
Looking for similar products? Visit Product Comparison Guide
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Lindsay R Comstock et al.
The Journal of organic chemistry, 69(4), 1425-1428 (2004-02-14)
8-Azido-5'-aziridino-5'-deoxyadenosine (6), a novel cofactor mimic, was synthesized in nine steps from commercially available 2',3'-isopropylideneadenosine in approximately 4% overall yield. Crucial to this success was a very unorthodox phthalimide cleavage procedure, C8 azidation prior to aziridination and late stage alkylation
I D Golovatskiĭ et al.
Ukrainskii biokhimicheskii zhurnal (1978), 61(2), 64-69 (1989-03-01)
Transformation and uptake of [8-14C]-adenosine and its synthetic analog 2',3'-O-isopropylideneadenosine was studied in Zajdel hepatoma cells and their homogenates. Uptake and deamination of adenosine and 2',3'-O-isopropylideneadenosine by Zajdel hepatoma cells proceed differently. A small part of adenosine is phosphorylated and
M Brignoni et al.
Journal of cell science, 108 ( Pt 5), 1931-1943 (1995-05-01)
Madin-Darby canine kidney and other epithelial cell lines (e.g. Caco-2, MCF-10A and MCF-7) develop intracellular vacuoles composed of apical membrane displaying microvilli (VACs) when impaired from forming normal cell-to-cell contacts. In a previous publication, we showed that VACs are rapidly
Pierangela Ciuffreda et al.
Nucleosides, nucleotides & nucleic acids, 26(10-12), 1311-1313 (2007-12-11)
2 ',3 '-Isopropylidene group can be used as a molecular scaffold for the introduction of modifications at 5 ' and 1 ' positions of adenosine and these modified nucleosides are used to evaluate the biocatalytic activity of adenosine and adenylate
I D Golovatskiĭ et al.
Ukrainskii biokhimicheskii zhurnal (1978), 58(3), 37-40 (1986-05-01)
Transformation of synthesized 2',3'-O-isopropylidene adenosine was studied in comparison with adenosine in rat liver homogenates. It is stated that 2',3'-O-isopropylidene adenosine is subjected to deamination similar to adenosine but less intensively. Due to deamination 2',3'-O-isopropylidene inosine is formed from 2',3'-O-isopropylidene
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service