ALD00374
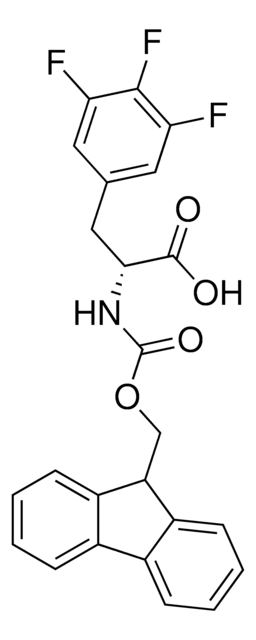
L-Fmoc-3-fluorophenylalanine
Synonym(s):
Fmoc-Phe(3-F)-OH, Fmoc-3-fluoro-L-phenylalanine
About This Item
Recommended Products
form
solid
Quality Level
reaction suitability
reaction type: Fmoc solid-phase peptide synthesis
color
beige
application(s)
peptide synthesis
functional group
Fmoc
storage temp.
2-8°C
SMILES string
FC1=CC=CC(C[C@H](NC(OCC2C3=C(C4=C2C=CC=C4)C=CC=C3)=O)C(O)=O)=C1
InChI
1S/C24H20FNO4/c25-16-7-5-6-15(12-16)13-22(23(27)28)26-24(29)30-14-21-19-10-3-1-8-17(19)18-9-2-4-11-20(18)21/h1-12,21-22H,13-14H2,(H,26,29)(H,27,28)/t22-/m0/s1
InChI key
DWSDVARCJDOADL-QFIPXVFZSA-N
Looking for similar products? Visit Product Comparison Guide
Application
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Sorry, we don't have COAs for this product available online at this time.
If you need assistance, please contact Customer Support.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Related Content
The Yu program centers around the discovery of catalytic carbon–carbon and carbon–heteroatom bond forming reactions based on C–H activation. Target transformations are selected to enable 1) the use of simple and abundant starting materials such as aliphatic acids, amines and alcohols, and 2) disconnections that drastically shorten the synthesis of a drug molecule or a major class of biologically active compounds.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service