All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C10H8FN3
CAS Number:
Molecular Weight:
189.19
UNSPSC Code:
12352106
NACRES:
NA.06
Recommended Products
Quality Level
Assay
≥95%
form
powder, crystals or chunks
color
light yellow
storage temp.
−20°C
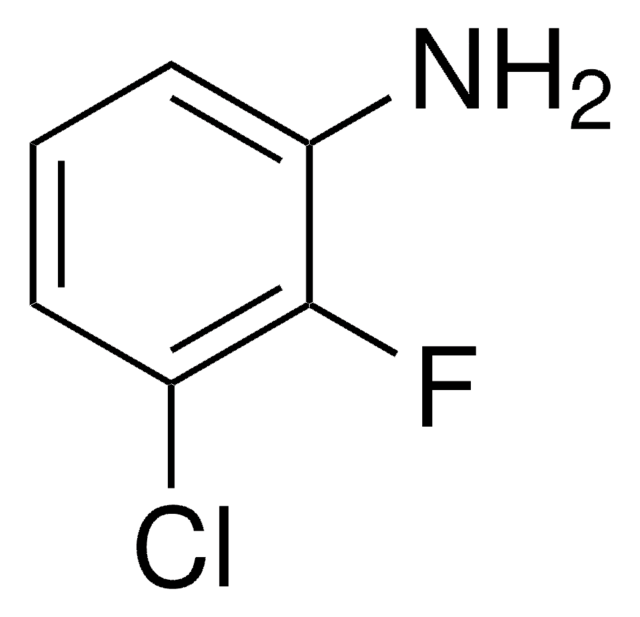
SMILES string
NC1=C(C2=CN=CN=C2)C=CC=C1F
Application
2-fluoro-6-(pyrimidin-5-yl)aniline is a temporary directing group (TDG) to assist as a co-catalyst for metal catalyzed C-H functionalization. Often in C-H functionalization, an auxiliary compound is used to control site selectivity. These traditionally are covalently bonded to the compound of interest, and must subsequently be removed after functionalization, like a typical protecting group. To simplify the process of C-H functionalization, 2-fluoro-6-(pyrimidin-5-yl)aniline is one of a series of temporary directing groups developed by Deb Maiti′s lab that promote site selectivity without the inclusion of additional synthetic steps.
2-fluoro-6-(pyrimidin-5-yl)aniline is an effective TDG for meta directed C-H functionalization of aldehyde substituted target compounds, with high selectivity.
2-fluoro-6-(pyrimidin-5-yl)aniline is an effective TDG for meta directed C-H functionalization of aldehyde substituted target compounds, with high selectivity.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Lot/Batch Number
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Sukdev Bag et al.
Nature communications, 12(1), 1393-1393 (2021-03-04)
Despite the widespread applications of C-H functionalization, controlling site selectivity remains a significant challenge. Covalently attached directing groups (DGs) served as ancillary ligands to ensure ortho-, meta- and para-C-H functionalization over the last two decades. These covalently linked DGs necessitate
Zhen Li et al.
Science (New York, N.Y.), 372(6549), 1452-1457 (2021-11-30)
Hydroxylation of aryl carbon-hydrogen bonds with transition metal catalysts has proven challenging when oxygen is used as the oxidant. Here, we report a palladium complex bearing a bidentate pyridine/pyridone ligand that efficiently catalyzes this reaction at ring positions adjacent to
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service


![Methyl {[(4-aminophenyl)acetyl]amino}acetate](/deepweb/assets/sigmaaldrich/product/structures/185/868/8f1e3cb5-81a3-4bbb-bc29-a8021071555f/640/8f1e3cb5-81a3-4bbb-bc29-a8021071555f.png)