All Photos(2)
About This Item
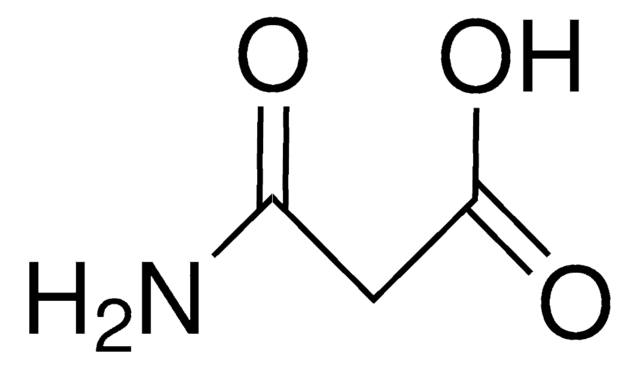
Linear Formula:
CH2(CONH2)2
CAS Number:
Molecular Weight:
102.09
Beilstein:
1751401
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
form
solid
mp
172-175 °C (lit.)
fluorescence
λex 367 nm; λem 445 nm (α-keto acid adduct)
SMILES string
NC(=O)CC(N)=O
InChI
1S/C3H6N2O2/c4-2(6)1-3(5)7/h1H2,(H2,4,6)(H2,5,7)
InChI key
WRIRWRKPLXCTFD-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
The malonamide derivatives are obtained by the one-pot, five-component condensation reaction of isocyanide, Meldrum′s acid, arylidene malononitrile, and two amine molecules in CH2Cl2.
Application
The malonamide-based ionic liquid extractant was used in the extraction of europium(iii) and other trivalent rare-earth ions from nitric acid medium.
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
L C Cerny et al.
Critical care medicine, 11(9), 739-743 (1983-09-01)
This study examines some of the biophysical properties of mixtures of whole blood with 3 different hydroxyethyl starch-hemoglobin (HES-Hgb) polymer blood substitutes. The polymers have about a 3-fold range of number average molecular weight and intrinsic viscosity range. The range
Abbas Rahmati et al.
Molecular diversity, 17(3), 619-625 (2013-05-25)
A one-pot, five-component condensation reaction of isocyanide, Meldrum's acid, arylidene malononitrile, and two amine molecules in CH2Cl2 at ambient temperature to give malonamide derivatives is described.
Bevin W Parks et al.
Inorganic chemistry, 45(4), 1498-1507 (2006-02-14)
This report describes an investigation into the coordination chemistry of trivalent lanthanides in solution and the solid state with acyclic and preorganized bicyclic malonamide ligands. Two experimental investigations were performed: solution binding affinities were determined through single-phase spectrophotometric titrations and
Romain Diss et al.
Physical chemistry chemical physics : PCCP, 7(2), 264-272 (2005-01-21)
According to molecular dynamics simulations, uncomplexed malonamide ligands L and their neutral Eu(NO3)3L2 or charged EuL4(3+) complexes are surface active and adsorb at a water-"oil" interface, where "oil" is modeled by chloroform. Aqueous solvation at the interface is found to
Alok Rout et al.
Journal of hazardous materials, 221-222, 62-67 (2012-05-01)
The extraction behavior of U(VI), Pu(IV) and Am(III) from nitric acid medium by a solution of N,N-dimethyl-N,N-dioctyl-2-(2-hexyloxyethyl)malonamide (DMDOHEMA) in the room temperature ionic liquid, 1-butyl-3-methylimidazolium bis(trifluoromethanesulfonyl)imide (C(4)mimNTf(2)), was studied. The distribution ratio of these actinides in DMDOHEMA/C(4)mimNTf(2) was measured as
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service