120227
Diethyl 1,4-dihydro-2,6-dimethyl-3,5-pyridinedicarboxylate
95%
Synonym(s):
Hantzsch ester
Sign Into View Organizational & Contract Pricing
All Photos(4)
About This Item
Empirical Formula (Hill Notation):
C13H19NO4
CAS Number:
Molecular Weight:
253.29
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
95%
form
solid
mp
178-183 °C (lit.)
solubility
organic solvents: soluble
functional group
ester
SMILES string
CCOC(=O)C1=C(C)NC(C)=C(C1)C(=O)OCC
InChI
1S/C13H19NO4/c1-5-17-12(15)10-7-11(13(16)18-6-2)9(4)14-8(10)3/h14H,5-7H2,1-4H3
InChI key
LJXTYJXBORAIHX-UHFFFAOYSA-N
Related Categories
General description
Diethyl 1,4-dihydro-2,6-dimethyl-3,5-pyridinedicarboxylate is often used as a building block in organic synthesis for the preparation of various biologically active compounds.
Application
Diethyl 1,4-dihydro-2,6-dimethyl-3,5-pyridinedicarboxylate (DTP) was used to study the mechanism of electrochemical oxidation of DTP in ethanol/water solutions on a glassy carbon electrode.
Used as a hydrogen source in organocatalytic reductive amination and conjugate reduction.
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
G I Klebanov et al.
Biomeditsinskaia khimiia, 52(1), 69-82 (2006-06-03)
Antioxidant activity (AA) of inhibitors of free radical reactions (FRR) (dieton, mexidol, trypsin), aplied to the dressing material for wound healing was studied. In our work we used a model system containing suspension of laminated liposome, formed from fraction of
V N Kovalenko et al.
Voprosy pitaniia, (2)(2), 44-49 (1982-03-01)
The efficacy of pharmacopeial alpha-tocopheryl acetate, alpha-tocopheronolactone and the antioxidant diludin was studied as to the possibilities of preventing E-hypovitaminosis muscle dystrophy in rabbits. alpha-Tocopheronolactone action was similar to that of pharmacopeial alpha-tocopheryl acetate as regards E-vitamin activity that was
N A Basova et al.
Rossiiskii fiziologicheskii zhurnal imeni I.M. Sechenova, 88(5), 650-657 (2002-07-26)
The main concern of this work was to examine the relation between altered antioxidant status on the one hand and increase in L-tryptophan absorption in the small intestine in order to bring further information regarding to possible role of vitamin
Jing Zhang et al.
iScience, 23(1), 100755-100755 (2019-12-31)
The alkoxyl radical is an essential reactive intermediate in mechanistic studies and organic synthesis with hydrogen atom transfer (HAT) reactivity. However, compared with intramolecular 1,5-HAT or intermolecular HAT of alkoxyl radicals, the intramolecular 1,2-HAT reactivity has been limited to theoretical
V F Dubrovskaja et al.
Radiobiologia, radiotherapia, 31(1), 79-84 (1990-01-01)
Female mice (CBA X C57 Bl) F1 were exposed a single total-body gamma-irradiation with a dose efficiency of 6.5 Gy/min with doses of 2.5-7.5 Gy in air or inhaling a gas mixture of 6-6.5% oxygen and 94-93% nitrogen. All naturally
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service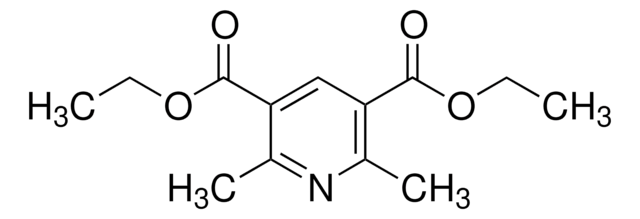








![[Ir(dtbbpy)(ppy)2]PF6](/deepweb/assets/sigmaaldrich/product/structures/158/329/2544d673-d267-4aa1-8f46-2652aad4bfa0/640/2544d673-d267-4aa1-8f46-2652aad4bfa0.png)
![Tris[2-phenylpyridinato-C2,N]iridium(III) sublimed grade](/deepweb/assets/sigmaaldrich/product/structures/167/234/658d0b76-d31d-4fd5-8041-e04e207227c9/640/658d0b76-d31d-4fd5-8041-e04e207227c9.png)