86320
Tartronic acid
≥97.0%
Synonym(s):
Hydroxymalonic acid, Hydroxypropanedioic acid
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
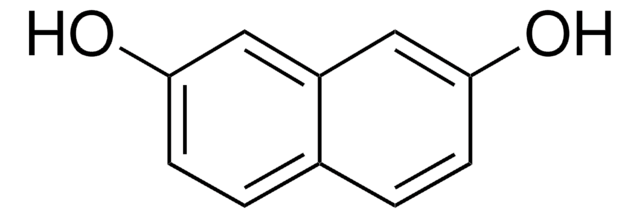
Linear Formula:
HOCH(COOH)2
CAS Number:
Molecular Weight:
120.06
Beilstein:
1209791
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
≥97.0%
form
powder
mp
158-160 °C (dec.) (lit.)
storage temp.
2-8°C
SMILES string
OC(C(O)=O)C(O)=O
InChI
1S/C3H4O5/c4-1(2(5)6)3(7)8/h1,4H,(H,5,6)(H,7,8)
InChI key
ROBFUDYVXSDBQM-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
- Polymer synthesis for enhanced thermal conductivity: Tartronic acid is used to exploit enzyme reactions in polymer synthesis, significantly increasing the thermal conductivity of materials, which is pivotal in manufacturing and material science applications (Nan et al., 2023).
- Advances in green chemical treatments: This acid plays a role in the electro-oxidation pathways for treating glycerol waste, contributing to sustainable chemical processes and green chemistry applications, which are essential for reducing environmental impact (Cheng et al., 2021).
- Development in biodiesel by-products treatment: Tartronic acid is also involved in kinetic studies for the electrochemical conversion of glycerol, a by-product of biodiesel production, highlighting its role in renewable energy and waste valorization (Pérès et al., 2020).
- Base-free oxidation reactions: It aids in the development of base-free conditions for glycerol to glyceraldehyde oxidation reactions over platinum-based catalysts, offering advancements in catalysis and organic synthesis processes (Capron et al., 2019).
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
J Klimek et al.
Journal of steroid biochemistry, 26(1), 161-163 (1987-01-01)
It has been shown that the conversion of cholesterol to progesterone by human term placental mitochondria incubated in the presence of malate or fumarate was inhibited by hydroxymalonate--an inhibitor of malic enzyme. No inhibition was observed when mitochondria were incubated
A Cortés et al.
The Biochemical journal, 357(Pt 1), 263-268 (2001-06-21)
The concentration of an inhibitor that decreases the rate of an enzyme-catalysed reaction by 50%, symbolized i(0.5), is often used in pharmacological studies to characterize inhibitors. It can be estimated from the common inhibition plots used in biochemistry by means
K F Petersen et al.
Metabolism: clinical and experimental, 44(11), 1380-1383 (1995-11-01)
The relative roles of pyruvate kinase and malic enzyme in substrate cycling between pyruvate and oxaloacetate were examined in perfused livers of 24-hour-fasted normal and triiodothyronine (T3)-treated rats using an inhibitor of malic enzyme (hydroxymalonate). Livers were perfused for 60
P A Tipton
Biochemistry, 35(9), 3108-3114 (1996-03-05)
The oxidative decarboxylation of D-malate catalyzed by tartrate dehydrogenase has been analyzed by transient-state kinetic methods and kinetic isotope effect measurements. The reaction time courses show a burst of NADH formation prior to the attainment of the steady-state velocity. The
[Not Available].
W K RIEBEN et al.
Helvetica physiologica et pharmacologica acta, 4(3), 52-52 (1946-01-01)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service