208713
Calpain-1, Human Erythrocytes
Calpain-1, Human Erythrocytes, is a native calpain-1. A heterodimeric cysteine proteinase with low Ca2+ requirement (EC₅₀ = 2 µM).
Synonym(s):
μ-Calpain
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Recommended Products
Quality Level
form
liquid
specific activity
≥1000 units/mg protein
manufacturer/tradename
Calbiochem®
storage condition
OK to freeze
avoid repeated freeze/thaw cycles
shipped in
wet ice
storage temp.
−70°C
General description
Native calpain-1 from human erythrocytes. Ca2+-dependent cysteine proteinase with low Ca2+ requirement (half-maximal activation = 2 µM). Participates in the ATP release reaction of platelets stimulated with thrombin.
Native calpain-1 from human erythrocytes. Ca2+-dependent heterodimeric cysteine proteinase with low Ca2+ requirement (EC50= 2 µM).
Packaging
Please refer to vial label for lot-specific concentration.
Warning
Toxicity: Harmful (C)
Unit Definition
One unit is defined as the amount of enzyme that will hydrolyze 1 pmol Suc-LLVY-AMC in 1 min, 25°C using the Calpain Activity Assay Kit, Fluorogenic (Cat. No. QIA120). Note: 1 caseinolytic unit = 1 fluorogenic unit.
Physical form
In 20 mM imidazole, 5 mM β-mercaptoethanol, 1 mM EDTA, 1 mM EGTA, 30% glycerol, pH 6.8.
Preparation Note
Prepared from blood that has been shown by certified tests to be negative for HBsAg and for antibodies to HIV and HCV.
Reconstitution
Following initial thaw, aliquot and freeze (-70°C).
Analysis Note
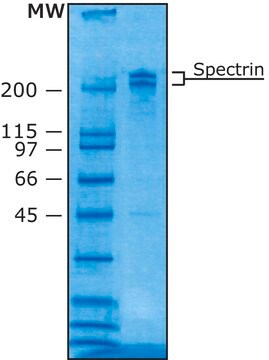
Comparable to reference lot by SDS-PAGE
Other Notes
Vanderklish, P.W., and Bahr, B.A. 2000. Int. J. Exp. Pathol.81, 323.
Sorimachi, H., et al. 1997. Biochem. J. 328, 721.
Croall, D.E., and McGrody, K.S. 1994. Biochemistry33, 13223.
Sorimachi, H., et al. 1997. Biochem. J. 328, 721.
Croall, D.E., and McGrody, K.S. 1994. Biochemistry33, 13223.
Legal Information
CALBIOCHEM is a registered trademark of Merck KGaA, Darmstadt, Germany
Storage Class Code
10 - Combustible liquids
WGK
WGK 2
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Eshwar R Tammineni et al.
eLife, 12 (2023-02-02)
Calcium ion movements between cellular stores and the cytosol govern muscle contraction, the most energy-consuming function in mammals, which confers skeletal myofibers a pivotal role in glycemia regulation. Chronic myoplasmic calcium elevation ("calcium stress"), found in malignant hyperthermia-susceptible (MHS) patients
Courtney Blachford et al.
Cell calcium, 46(4), 257-262 (2009-09-08)
Neuronal calcium sensor-1 (NCS-1) is a high-affinity, low-capacity Ca(2+)-binding protein expressed in many cell types. We previously showed that NCS-1 interacts with inositol 1,4,5-trisphosphate receptor (InsP(3)R) and modulates Ca(2+)-signaling by enhancing InsP3-dependent InsP(3)R channel activity and intracellular Ca(2+) transients. Recently
Michelle M White et al.
EBioMedicine, 23, 173-184 (2017-08-25)
Identification of mechanisms promoting neutrophil trafficking to the lungs of patients with cystic fibrosis (CF) is a challenge for next generation therapeutics. Cholesterol, a structural component of neutrophil plasma membranes influences cell adhesion, a key step in transmigration. The effect
BmATG5 and BmATG6 mediate apoptosis following autophagy induced by 20-hydroxyecdysone or starvation.
Kun Xie et al.
Autophagy, 12(2), 381-396 (2016-01-05)
Autophagy and apoptosis, which could be induced by common stimuli, play crucial roles in development and disease. The functional relationship between autophagy and apoptosis is complex, due to the dual effects of autophagy. In the Bombyx Bm-12 cells, 20-hydroxyecdysone (20E)
Peter Tompa et al.
The Journal of biological chemistry, 277(11), 9022-9026 (2002-01-26)
The inhibitory domains of calpastatin contain three highly conserved regions, A, B, and C, of which A and C bind calpain in a strictly Ca(2+)-dependent manner but have no inhibitory activity whereas region B inhibits calpain on its own. We
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service