G6104
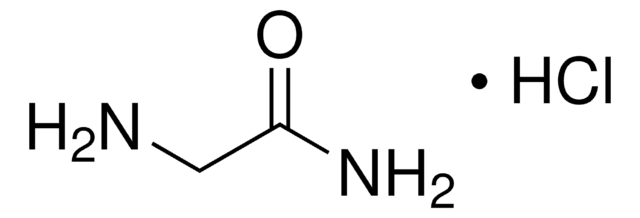
Glycinamide hydrochloride
98%
Synonyme(s) :
2-Aminoacetamide hydrochloride, Aminoacetamide hydrochloride, Glycine amide hydrochloride
Se connecterpour consulter vos tarifs contractuels et ceux de votre entreprise/organisme
About This Item
Formule linéaire :
NH2CH2CONH2 · HCl
Numéro CAS:
Poids moléculaire :
110.54
Numéro Beilstein :
3554199
Numéro CE :
Numéro MDL:
Code UNSPSC :
12352100
ID de substance PubChem :
Nomenclature NACRES :
NA.22
Produits recommandés
Niveau de qualité
Pureté
98%
Pf
204 °C (dec.) (lit.)
Chaîne SMILES
Cl.NCC(N)=O
InChI
1S/C2H6N2O.ClH/c3-1-2(4)5;/h1,3H2,(H2,4,5);1H
Clé InChI
WKNMKGVLOWGGOU-UHFFFAOYSA-N
Application
Buffer useful in the physiological pH range.
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
Eyeshields, Gloves, type N95 (US)
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Gottfried K Schroeder et al.
Biochemistry, 46(13), 4037-4044 (2007-03-14)
As a model for mechanistic comparison with peptidyl transfer within the ribosome, the reaction of aqueous glycinamide with N-formylphenylalanine trifluoroethyl ester (fPhe-TFE) represents an improvement over earlier model reactions involving Tris. The acidity of trifluoroethanol (pKa 12.4) resembles that of
Yong Sun et al.
The journal of physical chemistry. B, 109(12), 5919-5926 (2006-07-21)
For the purpose of investigating the tautomerism from glycinamide (G) to glycinamidic acid (G*) induced by proton transfer, we carried out a study of structural interconversion of the two tautomers and the relative stabilizing influences of water during the tautomerization
Irene M Lagoja et al.
Chemistry & biodiversity, 2(7), 923-927 (2006-12-29)
A possible reaction mechanism for the dehydration of glycinamide (3) and N,N'-diformylurea (4) yielding hypoxanthine (2) has been investigated. Furthermore, a potential prebiotic route converting hypoxanthine (2) into adenine (1) via phosphate activation followed by substitution reaction with NH3 was
Stefan Glatzel et al.
Chemical communications (Cambridge, England), 46(25), 4517-4519 (2010-05-21)
Homopolymers of N-acryloyl glycinamide were prepared by reversible addition-fragmentation chain transfer polymerization in water. The formed macromolecules exhibit strong polymer-polymer interactions in aqueous milieu and therefore form thermoreversible physical hydrogels in pure water, physiological buffer or cell medium.
Ajeet Singh et al.
Langmuir : the ACS journal of surfaces and colloids, 23(10), 5406-5411 (2007-04-14)
Conformational behaviors of urea and glycinamide have been investigated using the B3LYP functional with the 6-311+G* and 6-311+G** basis sets. Urea monomers have nonplanar minima at all the levels studied, even in the aqueous phase. In the case of glycinamide
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique