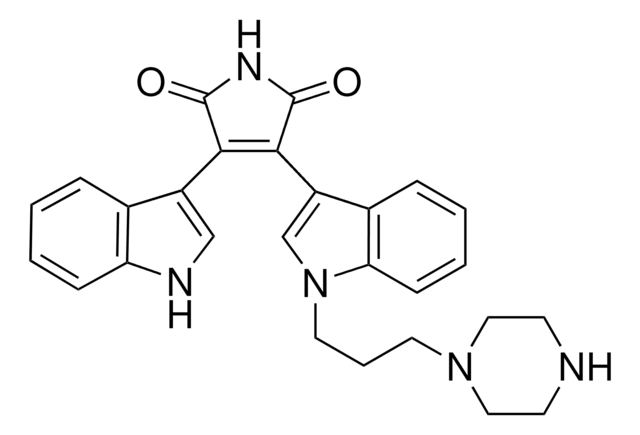
B3056
Bisindolylmaleimide II
≥97% (Mixture of 2 isomers)
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C27H26N4O2
CAS Number:
Molecular Weight:
438.52
MDL number:
UNSPSC Code:
12352111
PubChem Substance ID:
NACRES:
NA.77
Recommended Products
biological source
synthetic (organic)
Quality Level
Assay
≥97% (Mixture of 2 isomers)
form
powder
solubility
DMSO: soluble
ethanol: soluble
shipped in
dry ice
storage temp.
−20°C
SMILES string
CN1CCCC1CCn2cc(C3=C(C(=O)NC3=O)c4c[nH]c5ccccc45)c6ccccc26
InChI
1S/C27H26N4O2/c1-30-13-6-7-17(30)12-14-31-16-21(19-9-3-5-11-23(19)31)25-24(26(32)29-27(25)33)20-15-28-22-10-4-2-8-18(20)22/h2-5,8-11,15-17,28H,6-7,12-14H2,1H3,(H,29,32,33)
InChI key
LBFDERUQORUFIN-UHFFFAOYSA-N
Application
Bisindolylmaleimide II (BIS II) has been used:
- as a protein kinase C (PKC) inhibitor to study its effects on cardiomyocyte phenotypes
- as a PKC inhibitor along with methanandamide (mAEA) to study its effects on murine gastric vagal afferent (GVA) mechanosensitivity
- as a PKC/protein kinase A (PKA) inhibitor to study its effects on bone-marrow cells
Biochem/physiol Actions
Bisindolylmaleimide II (BIS II) acts as a dual protein kinase A/C (PKA/C) inhibitor.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Xiaojie Shi et al.
Brain research bulletin, 130, 138-145 (2017-01-25)
Carnosine is believed to be neuroprotective in cerebral ischemia. However, few reports concern its function on senescent astrocytes during cerebral ischemia. The aim of this study was to investigate the effects of carnosine on cell damage and glutamine synthetase (GS)
S E Wang et al.
Oncogene, 29(23), 3335-3348 (2010-04-13)
Activating mutations in the tyrosine kinase domain of HER2 (ErbB2) have been identified in human cancers. Compared with wild-type HER2, mutant HER2 shows constitutively activate kinase activity and increased oncogenicity. Cells transformed by mutant HER2 are resistant to epidermal growth
Binh Thanh Nguyen et al.
Electrophoresis, 37(23-24), 3146-3153 (2016-10-27)
An assay for protein kinase C delta (PKCδ) activity based on the quantification of a synthetic substrate using capillary electrophoresis with laser-induced fluorescence detection was developed. The peptides labeled with fluorescein isothiocyanate F-ERK (where ERK is extracellular signal-regulated kinase) and
Ameneh Cheshmehkani et al.
Biochemical pharmacology, 146, 139-150 (2017-09-26)
Agonism of the G protein-coupled free-fatty acid receptor-4 (FFA4) has been shown to promote numerous anti-inflammatory effects in macrophages that arise due to interaction with β-arrestin partner proteins. Humans express functionally distinct short and long FFA4 splice variants, such that
Salome Boroda et al.
Biochemical pharmacology, 123, 29-39 (2016-12-16)
Diacylglycerol kinase alpha (DGKα) catalyzes the conversion of diacylglycerol (DAG) to phosphatidic acid (PA). Recently, DGKα was identified as a therapeutic target in various cancers, as well as in immunotherapy. Application of small-molecule DGK inhibitors, R59022 and R59949, induces cancer
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service