38370
DIC
≥98.0% (GC), for peptide synthesis
Synonym(s):
N,N′-Diisopropylcarbodiimide
About This Item
Recommended Products
product name
DIC, purum, ≥98.0% (GC)
grade
purum
Quality Level
Assay
≥98.0% (GC)
form
liquid
reaction suitability
reaction type: Coupling Reactions
refractive index
n20/D 1.433 (lit.)
bp
145-148 °C (lit.)
density
0.815 g/mL at 20 °C (lit.)
0.815 g/mL at 20 °C
application(s)
peptide synthesis
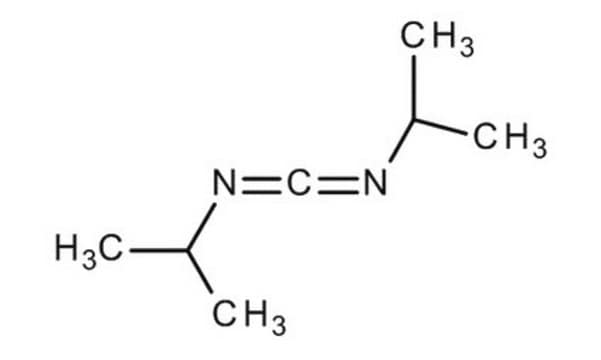
SMILES string
CC(C)N=C=NC(C)C
InChI
1S/C7H14N2/c1-6(2)8-5-9-7(3)4/h6-7H,1-4H3
InChI key
BDNKZNFMNDZQMI-UHFFFAOYSA-N
Gene Information
human ... EPHX2(2053)
mouse ... Ephx2(13850)
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
- To synthesize lanthanide (Ln) guanidinate complexes via insertion of carbodiimide into the Ln-N bond of lanthanocene secondary amido complexes.
- To facilitate the cyclization of N-(β-Hydroxy)amides to form 2-oxazolines.
- To synthesize 1-isopropyl-2-alkoxycarbonyl-3-isopropyliminio-aziridine by reacting with alkyl diazoacetates in the presence of transition metal salts.
- A coupling reagent for the synthesis of various esters and amides by treating carboxylic acids with phenols and amines respectively.
- A reagent for the conversion of alcohols to aldehydes or ketones in the presence of DMSO via modified Moffatt-type oxidation reaction.
- A reagent to facilitates the preparation of alkyl halides from corresponding alcohols via the formation of o-alkylisourea.
related product
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 1 Inhalation - Eye Dam. 1 - Flam. Liq. 3 - Resp. Sens. 1 - Skin Sens. 1
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
91.4 °F
Flash Point(C)
33 °C
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service