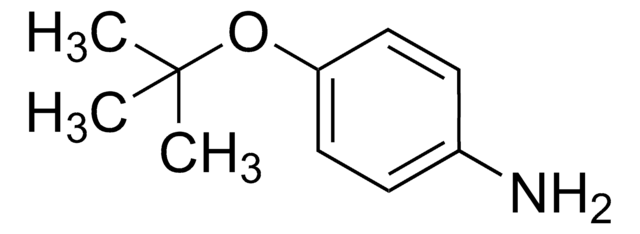
A88255
p-Anisidin
99%
Synonym(e):
4-Amino-anisol, 4-Methoxy-anilin
About This Item
Empfohlene Produkte
Qualitätsniveau
Assay
99%
Form
solid
Selbstzündungstemp.
959 °F
bp
240-243 °C (lit.)
mp (Schmelzpunkt)
56-59 °C (lit.)
SMILES String
COc1ccc(N)cc1
InChI
1S/C7H9NO/c1-9-7-4-2-6(8)3-5-7/h2-5H,8H2,1H3
InChIKey
BHAAPTBBJKJZER-UHFFFAOYSA-N
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Anwendung
p-Anisidine can be used:
- In the diastereoselective and enantioselective synthesis of CF3-substituted azoridines catalyzed by a chiral Bronsted acid.
- To prepare 4-organoselenium-quinolines through multi-component Povarov reaction with ethyl glyoxylate and ethynyl(phenyl)selane, catalyzed by Yb(OTf)3.
- As a starting material to synthesize 3-fluoro-6-methoxyquinoline in two steps.,
- To prepare N-PMP protected α-aminopropargylphosphonates by reacting with terminal alkynes and diethyl formylphosphonate hydrate using silver(I) triflate as a catalyst.
Signalwort
Danger
Gefahreneinstufungen
Acute Tox. 1 Dermal - Acute Tox. 2 Inhalation - Acute Tox. 2 Oral - Aquatic Acute 1 - Carc. 1B - STOT RE 2
Lagerklassenschlüssel
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
WGK
WGK 3
Flammpunkt (°F)
251.6 °F - closed cup
Flammpunkt (°C)
122 °C - closed cup
Hier finden Sie alle aktuellen Versionen:
Analysenzertifikate (COA)
Die passende Version wird nicht angezeigt?
Wenn Sie eine bestimmte Version benötigen, können Sie anhand der Lot- oder Chargennummer nach einem spezifischen Zertifikat suchen.
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.