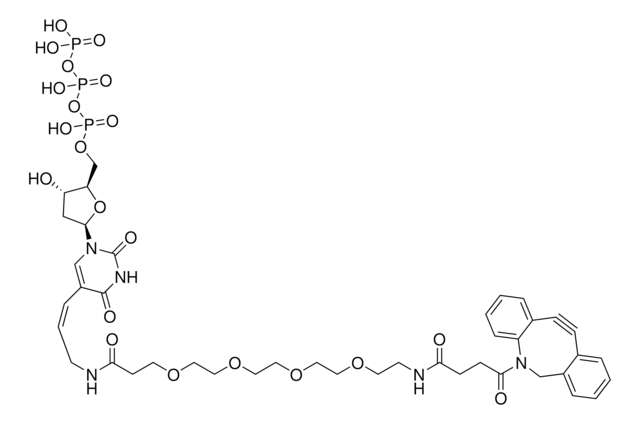
761540
Dibenzocyclooctyne-amine
for Copper-free Click Chemistry
Synonym(s):
DBCO-NH2, DBCO-amine
About This Item
Recommended Products
form
solid
reaction suitability
reaction type: click chemistry
reagent type: linker
mp
86-96 °C
functional group
amine
storage temp.
−20°C
SMILES string
NCCC(N1CC2=C(C=CC=C2)C#CC3=C1C=CC=C3)=O
InChI
1S/C18H16N2O/c19-12-11-18(21)20-13-16-7-2-1-5-14(16)9-10-15-6-3-4-8-17(15)20/h1-8H,11-13,19H2
InChI key
OCCYFTDHSHTFER-UHFFFAOYSA-N
Related Categories
General description
Application
- Linker to link azide-containing functional groups. It facilitates the formation of linkages through Strain-promoted azide-alkyne cycloaddition reactions (SPAAC)
- Reagent in the synthesis of N-heterocyclic carbene metal thiolates. It functionalizes the metal complexes and increases their reactivity in the strain-promoted alkyne–azide cycloaddition (SPAAC) reactions
- Amine functionalized cyclooctyne derivative. Cyclooctynes are useful in strain-promoted copper-free azide-alkyne cycloaddition reactions. This dibenzocyclooctyne will react with azide functionalized compounds or biomolecules without the need for a Cu(I) catalyst to result in a stable triazole linkage.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Copper-free click chemistry is an alternative approach to click chemistry that proceeds at a lower activation barrier and is free of cytotoxic transition metal catalysts.
Copper-free click chemistry is an alternative approach to click chemistry that proceeds at a lower activation barrier and is free of cytotoxic transition metal catalysts.
Copper-free click chemistry is an alternative approach to click chemistry that proceeds at a lower activation barrier and is free of cytotoxic transition metal catalysts.
Copper-free click chemistry is an alternative approach to click chemistry that proceeds at a lower activation barrier and is free of cytotoxic transition metal catalysts.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service



![(1R,8S,9s)-Bicyclo[6.1.0]non-4-yn-9-ylmethyl N-succinimidyl carbonate for Copper-free Click Chemistry](/deepweb/assets/sigmaaldrich/product/structures/969/022/d6776082-2f7a-47c7-bcd4-3830dac0fb7d/640/d6776082-2f7a-47c7-bcd4-3830dac0fb7d.png)
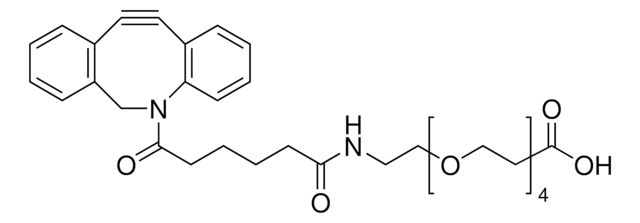
![(1R,8S,9s)-Bicyclo[6.1.0]non-4-yn-9-ylmethanol for Copper-free Click Chemistry](/deepweb/assets/sigmaaldrich/product/structures/171/632/0556139a-2db5-4678-a6ec-a26a693fd574/640/0556139a-2db5-4678-a6ec-a26a693fd574.png)

![N-[(1R,8S,9s)-Bicyclo[6.1.0]non-4-yn-9-ylmethyloxycarbonyl]-1,8-diamino-3,6-dioxaoctane for Copper-free Click Chemistry](/deepweb/assets/sigmaaldrich/product/structures/294/853/c5e47d84-5aee-4797-aa24-604f291171cc/640/c5e47d84-5aee-4797-aa24-604f291171cc.png)