C4733
BID, Caspase-8-cleaved from mouse
≥95% (SDS-PAGE), recombinant, expressed in E. coli, buffered aqueous solution
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Recommended Products
recombinant
expressed in E. coli
Quality Level
Assay
≥95% (SDS-PAGE)
form
buffered aqueous solution
UniProt accession no.
shipped in
dry ice
storage temp.
−20°C
Gene Information
mouse ... Bid(12122)
General description
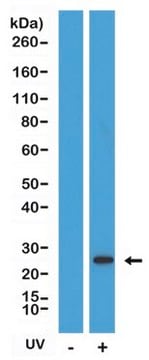
Mouse BID cleaved with caspase-8 generates the amino-terminal fragment (amino acids 1-59, 7 kDa) and the carboxy-terminal fragment (amino acids 60-195, 15 kDa). On size exclusion chromatography cleaved BID elutes at 27 kDa indicating that the fragments remain associated.
Biochem/physiol Actions
Caspase-8-cleaved BID relocates from the cytosol to the outer mitochondrial membrane where its interaction with Bak alters mitochondrial membrane permeability.
Physical form
0.2 μm filtered solution in 25 mM HEPES, pH 7.5, and 0.1 M KCl.
Analysis Note
Measured by its ability to induce cytochrome c release from isolated mouse liver mitochondria.
Storage Class Code
10 - Combustible liquids
WGK
nwg
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
J Zha et al.
Science (New York, N.Y.), 290(5497), 1761-1765 (2000-12-02)
Many apoptotic molecules relocate subcellularly in cells undergoing apoptosis. The pro-apoptotic protein BID underwent posttranslational (rather than classic cotranslational) N-myristoylation when cleavage by caspase 8 caused exposure of a glycine residue. N-myristoylation enabled the targeting of a complex of p7
M C Wei et al.
Genes & development, 14(16), 2060-2071 (2000-08-19)
TNFR1/Fas engagement results in the cleavage of cytosolic BID to truncated tBID, which translocates to mitochondria. Immunodepletion and gene disruption indicate BID is required for cytochrome c release. Surprisingly, the three-dimensional structure of this BH3 domain-only molecule revealed two hydrophobic
X Luo et al.
Cell, 94(4), 481-490 (1998-09-04)
We report here the purification of a cytosolic protein that induces cytochrome c release from mitochondria in response to caspase-8, the apical caspase activated by cell surface death receptors such as Fas and TNF. Peptide mass fingerprinting identified this protein
H Li et al.
Cell, 94(4), 491-501 (1998-09-04)
We report here that BID, a BH3 domain-containing proapoptotic Bcl2 family member, is a specific proximal substrate of Casp8 in the Fas apoptotic signaling pathway. While full-length BID is localized in cytosol, truncated BID (tBID) translocates to mitochondria and thus
BCL-2 family members and the mitochondria in apoptosis.
A Gross et al.
Genes & development, 13(15), 1899-1911 (1999-08-13)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service