Products may be shipped at a different temperature than the recommended long-term storage temperature. If the product quality is sensitive to short-term exposure to conditions other than the recommended long-term storage, it will be shipped on wet or dry-ice. If the product quality is NOT affected by short-term exposure to conditions other than the recommended long-term storage, it will be shipped at ambient temperature. As shipping routes are configured for minimum transit times, shipping at ambient temperature helps control shipping costs for our customers. For more information, please refer to the Storage and Transport Conditions document: https://www.sigmaaldrich.com/deepweb/assets/sigmaaldrich/marketing/global/documents/316/622/storage-transport-conditions-mk.pdf
P23954
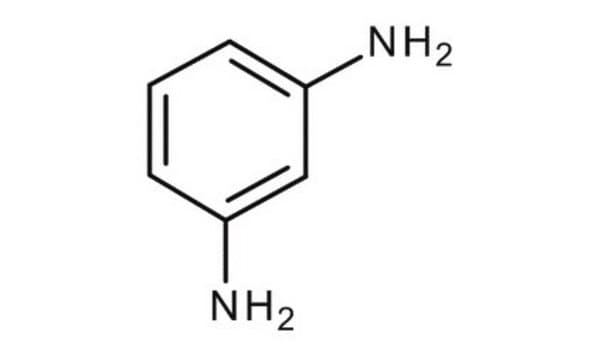
m-Phenylenediamine
flakes, 99%
Synonym(s):
1,3-Benzenediamine, 1,3-Diaminobenzene, 1,3-Phenylenediamine, MPDA
About This Item
Recommended Products
vapor density
3.7 (vs air)
Quality Level
vapor pressure
0.62 mmHg ( 100 °C)
Assay
99%
form
flakes
autoignition temp.
1040 °F
bp
282-284 °C
mp
64-66 °C
SMILES string
Nc1cccc(N)c1
InChI
1S/C6H8N2/c7-5-2-1-3-6(8)4-5/h1-4H,7-8H2
InChI key
WZCQRUWWHSTZEM-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
- intrinsically electrically semiconducting microparticles of semiladder poly(m-phenylenediamine-co-2-hydroxy-5-sulfonic aniline) structures[4]
- extraction medium based on chitosan-poly(m-phenylenediamine) (CS-PPD) Fe3O4 nanocomposite, used as sorbent for the magnetic solid-phase extraction (MSPE) of polychlorinated biphenyls[5]
- series of terpolymers, via chemical oxidative polymerization[6]
- thin film composite (TFC) membranes based polyamide[7]
- TFC reverse osmosis (RO) membranes[8]
Other Notes
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Eye Irrit. 2 - Muta. 2 - Skin Sens. 1
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
-
How is shipping temperature determined? And how is it related to the product storage temperature?
1 answer-
Helpful?
-
-
How can I determine the shelf life / expiration / retest date of this product?
1 answer-
If this product has an expiration or retest date, it will be shown on the Certificate of Analysis (COA, CofA). If there is no retest or expiration date listed on the product's COA, we do not have suitable stability data to determine a shelf life. For these products, the only date on the COA will be the release date; a retest, expiration, or use-by-date will not be displayed.
For all products, we recommend handling per defined conditions as printed in our product literature and website product descriptions. We recommend that products should be routinely inspected by customers to ensure they perform as expected.
For products without retest or expiration dates, our standard warranty of 1 year from the date of shipment is applicable.
For more information, please refer to the Product Dating Information document: https://www.sigmaaldrich.com/deepweb/assets/sigmaaldrich/marketing/global/documents/449/386/product-dating-information-mk.pdfHelpful?
-
-
What is the Department of Transportation shipping information for this product?
1 answer-
Transportation information can be found in Section 14 of the product's (M)SDS.To access the shipping information for this material, use the link on the product detail page for the product.
Helpful?
-
-
Does the purity of product P23954, m-Phenylendiamine, decrease substantially if it has darkened? Some batches of this product have been found to be white, and some are dark.
1 answer-
In most cases, the product is entirely usable after it has turned dark. Darkening may be apparent even when small amounts of product have been exposed to moisture in the atmosphere and there is no analyzable change in product purity. However, the products should not be used if a strong odor of ammonia is detected, as this indicates product degradation.The darkening is normal, and it is due to uptake of moisture from the atmosphere over time.
Helpful?
-
Active Filters
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service