393622
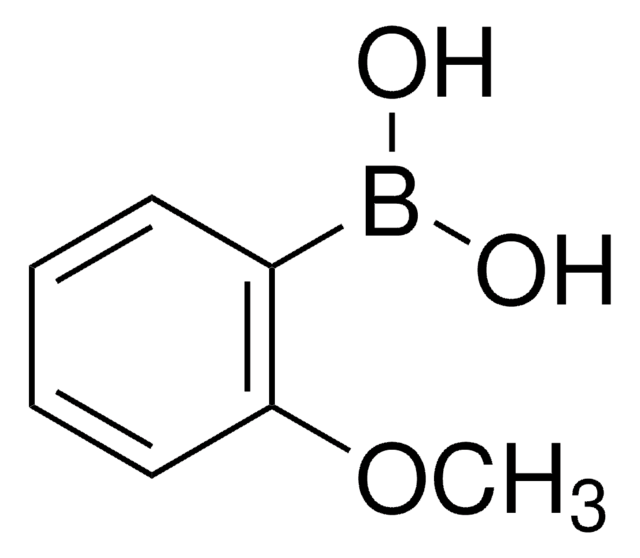
p-Tolylboronic acid
97%
Synonym(s):
(p-Methylphenyl)boronic acid, 4-Methylbenzeneboronic acid, 4-Methylphenylboronic acid, 4-Tolueneboronic acid, 4-Tolylboronic acid, p-Tolueneboronic acid, NSC 62870, p-Methylbenzeneboronic acid
About This Item
Recommended Products
Assay
97%
mp
256-263 °C (lit.)
SMILES string
Cc1ccc(cc1)B(O)O
InChI
1S/C7H9BO2/c1-6-2-4-7(5-3-6)8(9)10/h2-5,9-10H,1H3
InChI key
BIWQNIMLAISTBV-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
- Palladium (Pd)-catalyzed direct arylation
- Direct Palladium(II)-Catalyzed Synthesis
- Palladium-catalyzed arylation by Suzuki-Miyaura cross-coupling in water
- Cyclopalladation
- Tandem-type Pd(II)-catalyzed oxidative Heck reaction and intramolecular C-H amidation sequence
- Ruthenium catalyzed direct arylation
- Rhodium-catalyzed asymmetric conjugate addition
- Ligand-free copper-catalyzed cross-coupling reactions
- Regioselective arylation and alkynylation by Suzuki-Miyaura and Sonogashira cross-coupling reactions
- Ligand-free Suzuki, Sonogashira, and Heck cross-coupling reactions
Reagent used in Preparation of
- Catalysts for Suzuki-Miyaura cross-coupling of aryl bromides
- Recyclable Palladium nanoparticle catalysts immobilized by click ionic copolymers as for Suzuki-Miyaura cross-coupling reactions in water
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
MIDA-protected boronate esters offer stability, chromatography compatibility, and reactivity in anhydrous cross-coupling conditions.
MIDA-protected boronate esters offer stability, chromatography compatibility, and reactivity in anhydrous cross-coupling conditions.
MIDA-protected boronate esters offer stability, chromatography compatibility, and reactivity in anhydrous cross-coupling conditions.
MIDA-protected boronate esters offer stability, chromatography compatibility, and reactivity in anhydrous cross-coupling conditions.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service









![1-(4′-Methyl[1,1′-biphenyl]-4-yl)ethanone AldrichCPR](/deepweb/assets/sigmaaldrich/product/structures/224/118/064dfb31-1067-44ce-bf18-d3f762028eb6/640/064dfb31-1067-44ce-bf18-d3f762028eb6.png)
