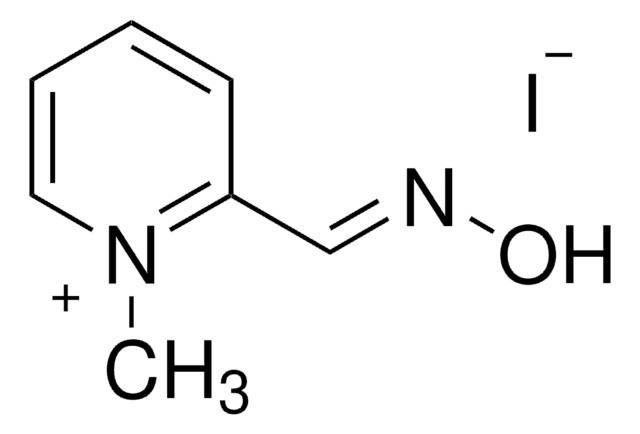
P9053
Pyridine-2-aldoxime methochloride
Synonym(s):
2-PAM chloride, Pralidoxime chloride
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Empirical Formula (Hill Notation):
C7H9N2O · Cl
CAS Number:
Molecular Weight:
172.61
EC Number:
MDL number:
UNSPSC Code:
12352202
PubChem Substance ID:
NACRES:
NA.77
Recommended Products
biological source
synthetic (organic)
Quality Level
form
solid
mp
230 °C (lit.)
solubility
water: 50 mg/mL, clear, colorless to faintly yellow
SMILES string
[Cl-].C[n+]1ccccc1\C=N\O
InChI
1S/C7H8N2O.ClH/c1-9-5-3-2-4-7(9)6-8-10;/h2-6H,1H3;1H
InChI key
HIGSLXSBYYMVKI-UHFFFAOYSA-N
Gene Information
human ... ACHE(43)
Looking for similar products? Visit Product Comparison Guide
Biochem/physiol Actions
The prototypical reactivator of acetylcholinesterase that has been inactivated by organophosphorus insecticides or nerve agents. It is now known that no reactivator is effective against a broad spectrum of organophosphorus agents.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Jarosław Kalisiak et al.
Journal of medicinal chemistry, 55(1), 465-474 (2011-12-31)
A new class of amidine-oxime reactivators of organophosphate (OP)-inhibited cholinesterases (ChE) was synthesized and tested in vitro and in vivo. Compared with 2-PAM, the most promising cyclic amidine-oxime (i.e., 12e) showed comparable or greater reactivation of OP-inactivated AChE and OP-inactivated
Kamil Musilek et al.
Bioorganic & medicinal chemistry letters, 17(11), 3172-3176 (2007-03-27)
Six novel AChE reactivators with a (Z)-but-2-ene linker were synthesized using the known synthetic pathways. Their ability to reactivate AChE, which had been previously inhibited by nerve agent tabun or pesticide paraoxon, was tested in vitro and compared to pralidoxime
F Worek et al.
Archives of toxicology, 72(4), 237-243 (1998-05-20)
The treatment of poisoning by highly toxic organophosphorus compounds (nerve agents) is unsatisfactory. Until now, the efficacy of new potential antidotes has primarily been evaluated in animals. However, the extrapolation of these results to humans is hampered by species differences.
Jyotiranjan Acharya et al.
European journal of medicinal chemistry, 44(3), 1335-1340 (2008-04-09)
A series of novel bis-pyridinium oximes connected by bis-methoxymethyl benzene, 1,4-bis-methoxymethyl (cis)-but-2-ene and 1,4-bis-methoxymethyl but-2-yne linkers were synthesized and their in vitro reactivation efficacy was evaluated against diisopropyl phosphorofluoridate (DFP) inhibited acetylcholinesterase (AChE) and compared with the established antidote 2-PAM
Jyotiranjan Acharya et al.
European journal of medicinal chemistry, 46(9), 3926-3933 (2011-06-28)
A series of carbamoyl bis-pyridinium monooximes linked with xylene linker were synthesized and their in-vitro reactivation potential was evaluated against acetylcholinesterase (AChE) inhibited by organophosphorus inhibitors (OP) such as sarin, DFP and VX and the data were compared with reactivation
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service