All Photos(1)
About This Item
Linear Formula:
BrC6H4HNCO2C(CH3)3
CAS Number:
Molecular Weight:
272.14
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
refractive index
n20/D 1.54 (lit.)
bp
110 °C/0.3 mmHg (lit.)
density
1.342 g/mL at 25 °C (lit.)
functional group
amine
bromo
SMILES string
CC(C)(C)OC(=O)Nc1ccccc1Br
InChI
1S/C11H14BrNO2/c1-11(2,3)15-10(14)13-9-7-5-4-6-8(9)12/h4-7H,1-3H3,(H,13,14)
InChI key
UQBGKDLSIIHUEZ-UHFFFAOYSA-N
General description
N-(tert-Butoxycarbonyl)-2-bromoaniline, also known as tert-butyl-N-(2-bromophenyl)carbamate, is an N-Boc-protected o-bromoaniline.1 It reacts with ethyl perfluorooctanoate in the presence of tert-butyllithium to form the corresponding 1-hydroxy-1H-perfluorooctyl compound.
Application
N-(tert-Butoxycarbonyl)-2-bromoaniline may be used in the synthesis of N-boc-protected o-aryl anilines, which undergoes coupling reaction with bromoacetylenes to form the corresponding arene?ynamides.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
230.0 °F - closed cup
Flash Point(C)
110 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Yousuke Yamaoka et al.
The Journal of organic chemistry, 80(2), 957-964 (2014-12-30)
A Brønsted acid-promoted arene-ynamide cyclization has been developed to construct the 3H-pyrrolo[2,3-c]quinolines. This reaction consists of the generation of a highly reactive keteniminium intermediate from arene-ynamide activated by a Brønsted acid and electrophilic aromatic substitution reaction to give arene-fused quinolines
Novel reduction of perfluoroalkyl ketones with lithium alkoxides.
Sokeirik YS, et al.
Journal of Fluorine Chemistry, 127(1), 150-152 (2006)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service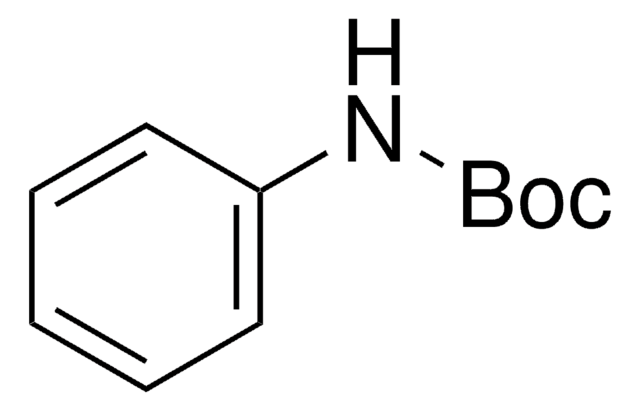
![4-[(N-Boc)aminomethyl]aniline 97%](/deepweb/assets/sigmaaldrich/product/structures/341/155/530c425c-7e6e-435e-a28a-9d40b05b938a/640/530c425c-7e6e-435e-a28a-9d40b05b938a.png)

![[4,4′-Bis(1,1-dimethylethyl)-2,2′-bipyridine] nickel (II) dichloride](/deepweb/assets/sigmaaldrich/product/structures/471/091/6faa29b1-bf8a-4d87-90b2-4cc55e082620/640/6faa29b1-bf8a-4d87-90b2-4cc55e082620.png)