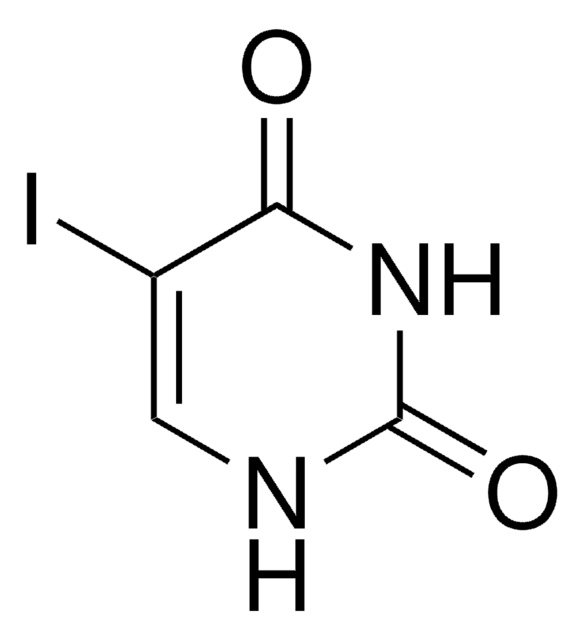
I6875
5-Iodocytosine
Synonym(s):
4-Amino-2-hydroxy-5-iodopyrimidine
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Empirical Formula (Hill Notation):
C4H4IN3O
CAS Number:
Molecular Weight:
237.00
MDL number:
UNSPSC Code:
41106305
PubChem Substance ID:
NACRES:
NA.51
Recommended Products
biological source
synthetic (organic)
Quality Level
Assay
≥99% (TLC)
form
powder
solubility
formic acid: 50 mg/mL
storage temp.
−20°C
SMILES string
[H]N1C=C(I)C(N)=NC1=O
InChI
1S/C4H4IN3O/c5-2-1-7-4(9)8-3(2)6/h1H,(H3,6,7,8,9)
InChI key
UFVWJVAMULFOMC-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
5-Iodocytosine is a modified pyrimidine used in the synthesis of molecules such as pyrrolocytosine and biologically active derivatives.
Application
5-Iodocytosine has been used as iodinated nucleotide with the single crystals of the 3D DNA designed motif for single anomalous dispersion (SAD) studies.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Chemistry for the synthesis of nucleobase-modified peptide nucleic acid
Hudson RHE, et al.
Pure and Applied Chemistry. Chimie Pure Et Appliquee, 76(7-8), 1591-1598 (2004)
T S Heuer et al.
Biochemistry, 36(35), 10655-10665 (1997-09-02)
The virally encoded integrase protein carries out retroviral integration, and to do so, it must make specific interactions with both viral and target DNA sequences. The retroviral integrase has three domains: an amino-terminal region of about 50 amino acids that
Y Yoshimura et al.
Bioorganic & medicinal chemistry, 8(7), 1545-1558 (2000-09-08)
As part of our ongoing investigation of the synthesis of biologically interesting 2'-modified-4'-thionucleosides, we synthesized 2'-deoxy-2'-fluoro-4'-thioarabinofuranosylpyrimidine and -purine nucleosides, and evaluated their antiviral and antitumor activities. In the pyrimidine series, beta-anomers of 5-ethyluracil, 5-iodouracil, 5-chloroethyluracil, and 5-iodocytosine derivatives showed potent
R H E Hudson et al.
Nucleosides, nucleotides & nucleic acids, 24(5-7), 581-584 (2005-10-27)
We have employed a tandem Sonogashira/annulation reaction between 5-iodocytosine derivatives and terminal alkynes to yield the fluorescent bicyclic nucleobase pyrrolcytosine. Pyrrolocytosine bearing substituents only on the pyrrole ring are conveniently synthesized from 5-iodocytosine. Water soluble pyrrolocytosines are being investigated as
S Harinipriya et al.
Journal of colloid and interface science, 250(1), 201-212 (2005-11-18)
The two-dimensional condensation behavior exhibited by nucleobases at a mercury/aqueous solution interface is analyzed on the basis of their hydrogen-bonded quadruplex structures, and the experimentally observed critical temperatures are rationalized incorporating different types of hydrogen bonding, surface coordination effects, and
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service