M19201
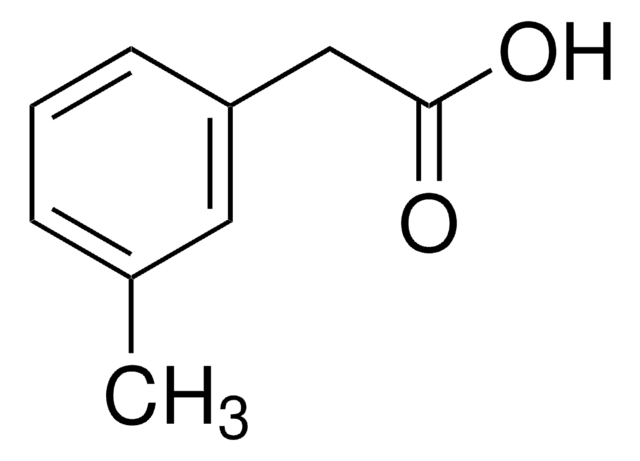
4-Methoxyphenylacetic acid
ReagentPlus®, 99%
Synonym(s):
Homoanisic acid
Sign Into View Organizational & Contract Pricing
All Photos(4)
About This Item
Linear Formula:
CH3OC6H4CH2CO2H
CAS Number:
Molecular Weight:
166.17
Beilstein:
1101737
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
product line
ReagentPlus®
Assay
99%
bp
140 °C/3 mmHg (lit.)
mp
84-86 °C (lit.)
SMILES string
COc1ccc(CC(O)=O)cc1
InChI
1S/C9H10O3/c1-12-8-4-2-7(3-5-8)6-9(10)11/h2-5H,6H2,1H3,(H,10,11)
InChI key
NRPFNQUDKRYCNX-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
4-Methoxyphenylacetic acid can be used:
- To prepare methyl 4-methoxyphenylacetate by esterification with dimethyl carbonate using mesoporous sulfated zirconia catalyst.
- As a ligand to synthesize pharmacologically important dinuclear gallium(III) and phenyltin(IV) carboxylate metal complexes.
- As a reactant to synthesize hydroxylated (E)-stilbenes by reacting with substituted benzaldehydes via Perkin reaction.
Legal Information
ReagentPlus is a registered trademark of Merck KGaA, Darmstadt, Germany
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Dam. 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
379.4 °F
Flash Point(C)
193 °C
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Esterification of 4-methoxyphenylacetic acid with dimethyl carbonate over mesoporous sulfated zirconia
Devulapelli VG and Weng H-S
Catalysis Communications, 10(13), 1711-1717 (2009)
An unusual, mild and convenient one-pot two-step access to (E)-stilbenes from hydroxy-substituted benzaldehydes and phenylacetic acids under microwave activation: a new facet of the classical Perkin reaction
Sinha AK, et al.
Tetrahedron, 63(45), 11070-11077 (2007)
Milena R Kaluderović et al.
European journal of medicinal chemistry, 45(2), 519-525 (2009-11-21)
The reaction of 3-methoxyphenylacetic acid, 4-methoxyphenylacetic acid, mesitylthioacetic acid, 2,5-dimethyl-3-furoic acid and 1,4-benzodioxane-6-carboxylic acid with trimethylgallium (1:1) yielded the dimeric complexes [Me(2)Ga(micro-O(2)CCH(2)C(6)H(4)-3-OMe)](2) (1), [Me(2)Ga(micro-O(2)CCH(2)C(6)H(4)-4-OMe)](2) (2), [Me(2)Ga(micro-O(2)CCH(2)SMes)](2) (3) (Mes=2,4,6-Me(3)C(6)H(2)), [Me(2)Ga{micro-O(2)C(Fur)}](2) (4) (Fur=2,5-dimethylfuran) and [Me(2)Ga{micro-O(2)C(Bdo)}](2) (5) (Bdo=1,4-benzodioxane) respectively. The molecular structure of
Santiago Gómez-Ruiz et al.
Journal of inorganic biochemistry, 102(12), 2087-2096 (2008-09-02)
The reaction of 3-methoxyphenylacetic acid (3-MPAH), 4-methoxyphenylacetic acid (4-MPAH), 2,5-dimethyl-3-furoic acid (DMFUH) or 1,4-benzodioxane-6-carboxylic acid (BZDOH) with triphenyltin(IV) chloride (1:1) or diphenyltin(IV) dichloride (2:1) in the presence of triethylamine yielded the compounds [SnPh3(3-MPA)] (1), [SnPh3(4-MPA)] (2), [SnPh3(DMFU)] (3), [SnPh3(BZDO)] (4)
Ki Hyun Kim et al.
Bioorganic & medicinal chemistry letters, 21(8), 2270-2273 (2011-03-23)
A bioassay-guided fractionation and chemical investigation of the trunk of Berberis koreana resulted in the isolation and identification of a new sesquilignan, named berbikonol (1), along with fourteen known lignan derivatives (2-15) and a new phenolic compound, named berfussinol (16)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service