All Photos(3)
About This Item
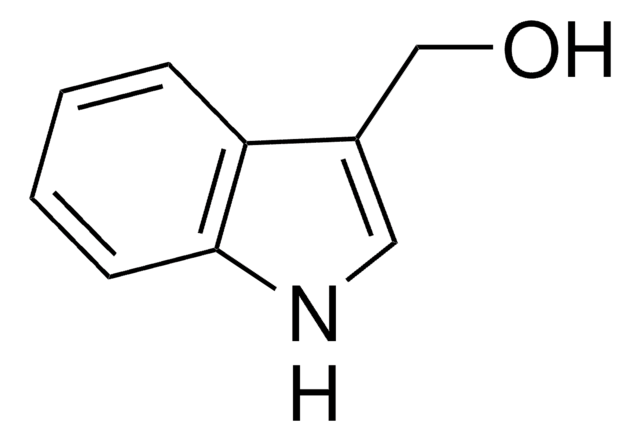
Empirical Formula (Hill Notation):
C8H7N
CAS Number:
Molecular Weight:
117.15
Beilstein:
107693
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
≥99%
bp
253-254 °C (lit.)
mp
51-54 °C (lit.)
SMILES string
c1ccc2[nH]ccc2c1
InChI
1S/C8H7N/c1-2-4-8-7(3-1)5-6-9-8/h1-6,9H
InChI key
SIKJAQJRHWYJAI-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
- Indole is a nitrogen-containing heterocycle used in the total synthesis of compounds such as goniomitine, (−)-isatisine A, and (±)-aspidospermidine.
- It is used as an electron donor moiety in synthesizing dyes for organic photovoltaics.
- It can also be used in the preparation of indole based conjugated small molecules for nonlinear optics applications.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 4 Oral - Aquatic Acute 1 - Eye Irrit. 2
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 1
Flash Point(F)
249.8 °F - closed cup
Flash Point(C)
121 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Synthesis and nonlinear optical properties of novel conjugated small molecules based on indole donor
Liu J, et al.
Journal of Molecular Structure, 1165(30), 223-227 (2018)
Impact of Thermal Annealing on Organic Photovoltaic Cells Using Regioisomeric Donor-Acceptor-Acceptor Molecules
Zhang T, et al.
ACS Applied Materials & Interfaces, 9(30), 25418-25425 (2017)
Catalytic selective cyclizations of aminocyclopropanes: formal synthesis of aspidospermidine and total synthesis of goniomitine
De Simone F, et al.
Angewandte Chemie (International ed. in English), 122(33), 5903-5906 (2010)
Regioselective Inter-and Intramolecular Formal [4+ 2] Cycloaddition of Cyclobutanones with Indoles and Total Synthesis of (?)-Aspidospermidine
Kawano M, et al.
Angewandte Chemie (International ed. in English), 125(3), 940-944 (2013)
Total Synthesis of (−)-Isatisine A
Zhang X, et al.
Angewandte Chemie (International ed. in English), 50(27), 6164-6166 (2011)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service