465151
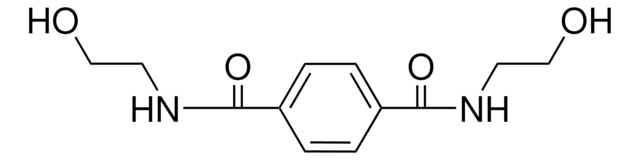
Bis(2-hydroxyethyl) terephthalate
Synonym(s):
Bis(β-hydroxyethyl) terephthalate, Bis(2-hydroxyethyl) terephthalate, Bis(ethylene glycol) terephthalate, Bis(hydroxyethyl) terephthalate, Terephthalic acid diethylene glycol ester
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Linear Formula:
C6H4-1,4-(CO2CH2CH2OH)2
CAS Number:
Molecular Weight:
254.24
EC Number:
MDL number:
UNSPSC Code:
12162002
PubChem Substance ID:
NACRES:
NA.23
Recommended Products
form
solid
Quality Level
mp
106-109 °C (lit.)
SMILES string
OCCOC(=O)c1ccc(cc1)C(=O)OCCO
InChI
1S/C12H14O6/c13-5-7-17-11(15)9-1-2-10(4-3-9)12(16)18-8-6-14/h1-4,13-14H,5-8H2
InChI key
QPKOBORKPHRBPS-UHFFFAOYSA-N
Related Categories
General description
Bis(2-hydroxyethyl) terephthalate belongs to the class of monomers known as diols or glycols. It is widely used to synthesize unsaturated polyester resins, polyethylene terephthalate (PET), and new biocompatible polymer systems. BHET can also be used as a plasticizer in polyester resins and rigid or flexible polyurethanes. Additionally, BHET and its derivatives have also been investigated for their potential use in biomedical applications, including drug delivery and tissue engineering, due to their excellent biocompatibility and mechanical properties.
Application
Bis(2-hydroxyethyl) terephthalate canbe used as a monomer to prepare biodegradable aromatic-aliphatic copolyestersfor sustainable flexible packaging applications. The incorporation of BHET as abuilding block offers a promising approach to enhancing the properties ofbiodegradable plastics and promoting sustainable packaging solutions.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Yanbo Zhang et al.
International journal of biological macromolecules, 166, 251-258 (2020-10-31)
Hydrothermal degradation was used to pretreat terylene with an aim of noticeably improving the yield of fermentable monomers: terephthalic acid (TPA), mono (2- hydroxyethyl) terephthalic acid (MHET), bis-hydroxyethyl terephthalate (BHET), and ethylene glycol (EG). After 0.5 h of reaction time at
Martin Eckardt et al.
Food additives & contaminants. Part A, Chemistry, analysis, control, exposure & risk assessment, 36(12), 1882-1894 (2019-09-04)
Linear and cyclic oligomers are unavoidable non-intentionally added substances (NIAS) present in food contact materials made from common polyesters such as polyethylene terephthalate (PET), polybutylene terephthalate (PBT) and polyester coatings. Although polyester oligomers can migrate into fats in significant amounts
Study on depolymerization of waste polyethylene terephthalate into monomer of bis(2-hydroxyethyl terephthalate
Guoxi Xi, et al.
Polymer Degradation and Stability, 87, 117-120 (2005)
Makoto Furukawa et al.
ChemSusChem, 11(23), 4018-4025 (2018-10-07)
Enzymatic degradation of poly(ethylene terephthalate) (PET) is promising because this process is safer than conventional industrial approaches. Recently, a cationic PET hydrolase (PETase) was identified from Ideonella sakaiensis. Pre-incubation of a low-crystallinity PET film with anionic surfactants prior to initiating
Uschara Thumarat et al.
Journal of bioscience and bioengineering, 120(5), 491-497 (2015-04-26)
This study described the genetic map of tandem genes (est1 and est119) encoding cutinase-type polyesterases in Thermobifida alba AHK119 and comparison of wild type and mutant enzymes of Est1 and Est119. Two genes were independently and constitutively expressed. The activity
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service