All Photos(2)
About This Item
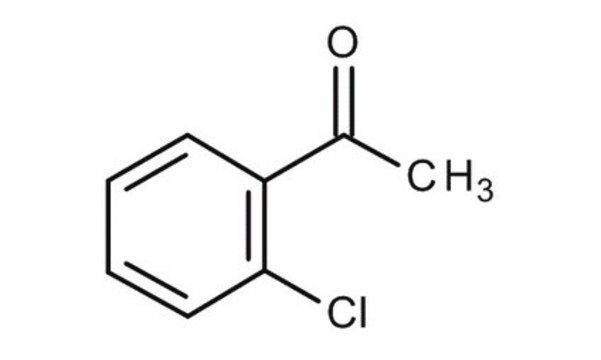
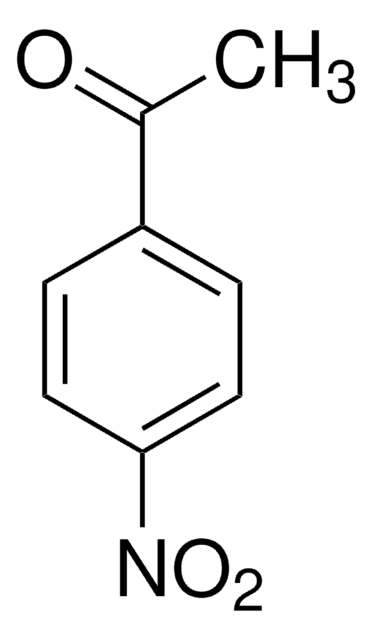
Linear Formula:
ClC6H4COCH3
CAS Number:
Molecular Weight:
154.59
Beilstein:
1858916
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
form
liquid
bp
227-230 °C
density
1.190 g/mL at 20 °C
functional group
chloro
ketone
SMILES string
CC(=O)c1ccccc1Cl
InChI
1S/C8H7ClO/c1-6(10)7-4-2-3-5-8(7)9/h2-5H,1H3
InChI key
ZDOYHCIRUPHUHN-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
2′-Chloroacetophenone undergoes stereoselective reduction to (R)-2′-chloro-1-phenyl-ethanol by Saccharomyces cerevisiae B5. It is commonly used as lacrimator.
Application
2′-Chloroacetophenone was employed as model substrate to investigate the enzymatic performance of Aspergillus terreus and Rhizopus oryzae in enantioselective bioreductions using glycerol as a co-solvent.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
WGK
WGK 3
Flash Point(F)
190.4 °F
Flash Point(C)
88 °C
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
K E Ferslew et al.
Journal of forensic sciences, 31(2), 658-665 (1986-04-01)
2-Chloroacetophenone (CN) and o-chlorobenzylidene malononitrile (CS) are the most common chemical agents used as lacrimators in the United States. There is a lack of complete spectral data on these compounds in the literature. Spectral data (ultraviolet, fluorescence, proton nuclear magnetic
Hui Song et al.
Journal of chromatographic science, 57(4), 299-304 (2019-02-06)
The synthetic condition of tulobuterol was optimized to gain lower impurity content. Two intermediates were analyzed, and three degradation impurities were isolated using preparative liquid chromatography for the first time and subsequently characterized by various techniques. Possible degradation impurities were
Improving the enantioselective bioreduction of aromatic ketones mediated by Aspergillus terreus and Rhizopus oryzae: the role of glycerol as a co-solvent.
Andrade LH, et al.
Tetrahedron Asymmetry, 20(13), 1521-1525 (2009)
Brigitte Holzer et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 26(13), 2869-2882 (2019-11-16)
A reliable synthetic protocol toward a series of fused chalcogenopheno[1]benzochalcogenophene (CBC) building blocks was developed based on a Fiesselmann reaction. The obtained CBC units were applied in McMurry and Stille coupling reactions toward symmetric regioisomeric ene-linked dimers. These π-conjugated compounds
Zhi-Min Ou et al.
Sheng wu gong cheng xue bao = Chinese journal of biotechnology, 19(2), 206-211 (2005-06-22)
(R)-chlorprenaline, a selective activator of beta2 receptor and an effective drug for bronchitis and asthma, is industrially prepared from (R)-2'-chloro-1-phenyl-ethanol. In this communication, we describe (1) the identification of Saccharomyces cerevisiae B5 as an effective host for stereoselective reduction of
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service