P45605
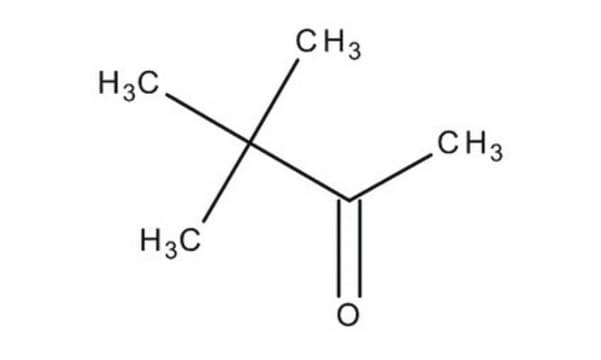
3,3-Dimethyl-2-butanone
97%
Synonym(s):
α,α,α-Trimethylacetone, tert-Butyl methyl ketone, Pinacolone
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
CH3COC(CH3)3
CAS Number:
Molecular Weight:
100.16
Beilstein:
1209331
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
refractive index
n20/D 1.396 (lit.)
bp
106 °C (lit.)
density
0.801 g/mL at 25 °C (lit.)
SMILES string
CC(=O)C(C)(C)C
InChI
1S/C6H12O/c1-5(7)6(2,3)4/h1-4H3
InChI key
PJGSXYOJTGTZAV-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
3,3-Dimethyl-2-butanone is an aliphatic ketone can undergo asymmetric reduction to the corresponding alcohol with diisopinocampheylchloroborane with high enantiomeric excess.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Flam. Liq. 2
Storage Class Code
3 - Flammable liquids
WGK
WGK 2
Flash Point(F)
41.0 °F - closed cup
Flash Point(C)
5 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Highly efficient asymmetric reduction of. alpha.-tertiary alkyl ketones with diisopinocampheylchloroborane.
Brown H C, et al.
The Journal of Organic Chemistry, 51(17), 3394-3396 (1986)
Kosuke Namba et al.
Organic letters, 14(5), 1222-1225 (2012-02-22)
An efficient method for the construction of dihydroquinoline derivatives possessing a quaternary carbon center is developed by an application of Hg(OTf)(2)-catalyzed vinylogous semi-pinacol-type rearrangement. The reaction was found to be specifically catalyzed by mercury salt and to proceed via a
Y Chen et al.
The Journal of organic chemistry, 66(11), 3930-3939 (2001-05-26)
To investigate the effects of electron-donating and electron-withdrawing substituents upon the reaction of porphyrins with osmium tetraoxide, and the pinacol-pinacolone rearrangement of the resulting diols, a series of meso-substituted porphyrins were prepared by total synthesis. Porphyrins with electron-donating substitutents at
Keliang Gao et al.
Applied microbiology and biotechnology, 71(6), 819-823 (2006-02-21)
Enantioselective biotransformation of DL-1,2-propanediol to D-2-hydroxypropanic acid was first reported by the authors. In the biooxidation process, there were some by-product formed and thus influenced the e.e. value and output of the acid. Restricting oxygen in the reaction system and
Makoto Shimizu et al.
Organic letters, 4(23), 4097-4099 (2002-11-09)
The pinacol reaction of beta-halogenated alpha,beta-unsaturated aldehydes was promoted by titanium tetraiodide to give coupling products in good yields with high dl-selectivity. Subsequent reduction with H(2)/Pd-C gave saturated vic-diols in good yields. Heck coupling reaction enabled the displacement of halogens
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service