93690
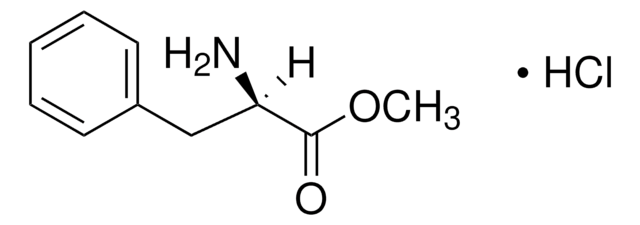
L-Tryptophan ethyl ester hydrochloride
≥99.0% (AT)
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C13H16N2O2 · HCl
CAS Number:
Molecular Weight:
268.74
Beilstein:
3919010
EC Number:
MDL number:
UNSPSC Code:
12352209
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
≥99.0% (AT)
form
powder
optical activity
[α]20/D +10±1°, c = 2% in H2O
reaction suitability
reaction type: solution phase peptide synthesis
mp
220-225 °C (dec.)
application(s)
peptide synthesis
SMILES string
Cl.CCOC(=O)[C@@H](N)Cc1c[nH]c2ccccc12
InChI
1S/C13H16N2O2.ClH/c1-2-17-13(16)11(14)7-9-8-15-12-6-4-3-5-10(9)12;/h3-6,8,11,15H,2,7,14H2,1H3;1H/t11-;/m0./s1
InChI key
PESYCVVSLYSXAK-MERQFXBCSA-N
Looking for similar products? Visit Product Comparison Guide
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Kento Takayama et al.
Journal of natural medicines, 75(1), 116-128 (2020-10-21)
Indole is produced from dietary tryptophan by tryptophanase in intestinal bacteria, such as Escherichia coli. In the liver, indole is converted into indoxyl sulfate, a uremic toxin and risk factor for chronic kidney disease (CKD). Probiotics and prebiotics are currently
Diana E Schlamadinger et al.
The journal of physical chemistry. B, 113(44), 14769-14778 (2009-10-13)
Ultraviolet resonance Raman (UVRR) spectra of tryptophan compounds in various solvents and a model peptide are presented and reveal systematic changes that reflect solvent polarity, hydrogen bond strength, and cation-pi interaction. The commonly utilized UVRR spectral marker for environment polarity
V Iu Shviadas et al.
Biokhimiia (Moscow, Russia), 45(4), 629-635 (1980-04-01)
The hydrolysis of L-tryptophane ethyl ester catalyzed by alpha-chymotrypsin and the effect of ethyl ster of D-tryptophane on the course of the reaction were studied. A kinetic pattern of a three-step enzymatic reaction based on the assumption that the enzyme
F L Tse et al.
The Journal of pharmacy and pharmacology, 36(9), 633-636 (1984-09-01)
Sandoz compound 57-118 is a mixture of tryptophan ethyl ester amide derivatives (analogues I-V) possessing one of five fatty acid chains which differ in chain length, configuration, or the degree of unsaturation. The relative absorption of each of the five
F Kilár et al.
Electrophoresis, 16(8), 1510-1518 (1995-08-01)
Enantiomers can be separated by using human serum transferrin as a chiral phase. With the help of the native protein we were able to separate enantiomers with high efficiency, using a low ionic strength 2-(N-morpholino)ethanesulfonic acid (MES) buffer, pH 6
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service