I0060000
Ifosfamide
European Pharmacopoeia (EP) Reference Standard
동의어(들):
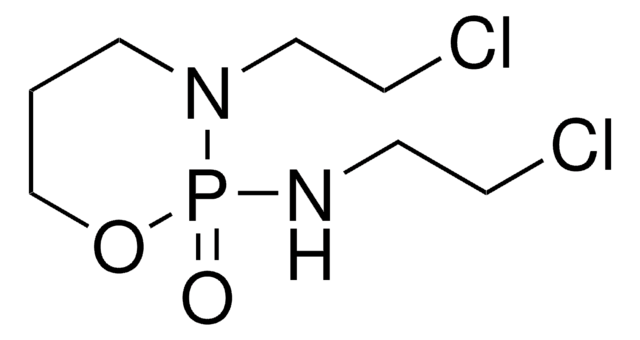
Ifex, N,3-Bis(2-chloroethyl)tetrahydro-2H-1,3,2-oxazaphosphorin-2-amine-2-oxide
About This Item
추천 제품
Grade
pharmaceutical primary standard
API family
ifosfamide
제조업체/상표
EDQM
기술
HPLC: suitable
gas chromatography (GC): suitable
응용 분야
pharmaceutical (small molecule)
형식
neat
저장 온도
2-8°C
SMILES string
ClCCNP1(=O)OCCCN1CCCl
InChI
1S/C7H15Cl2N2O2P/c8-2-4-10-14(12)11(6-3-9)5-1-7-13-14/h1-7H2,(H,10,12)
InChI key
HOMGKSMUEGBAAB-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
애플리케이션
생화학적/생리학적 작용
포장
기타 정보
관련 제품
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 3 Oral - Carc. 1B - Eye Irrit. 2 - Muta. 1B - Repr. 1B
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
Choose from one of the most recent versions:
시험 성적서(COA)
Sorry, we don't have COAs for this product available online at this time.
If you need assistance, please contact 고객 지원 부서
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.