추천 제품
분석
≥97.5% (GC)
97.5-102.5% (T)
형태
solid
적합성
complies for identity (IR)
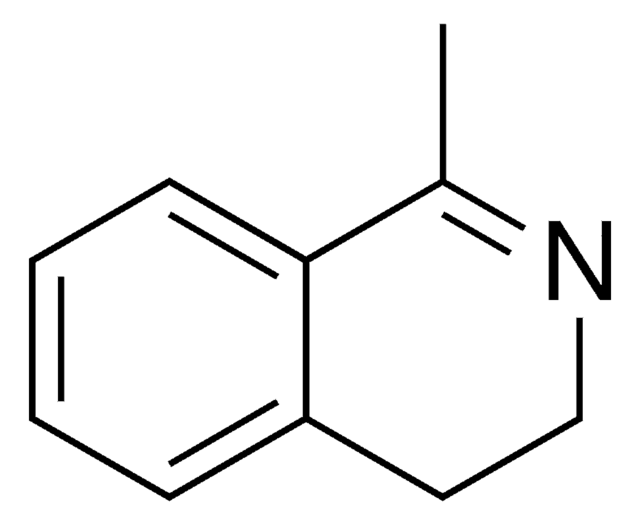
SMILES string
C1Cc2ccccc2C=N1
InChI
1S/C9H9N/c1-2-4-9-7-10-6-5-8(9)3-1/h1-4,7H,5-6H2
InChI key
NKSZCPBUWGZONP-UHFFFAOYSA-N
애플리케이션
3,4-Dihydroisoquinoline can be used as a reactant to synthesize:
- 5,6-Dihydro-8H-isoquino[1,2-b]quinazolin-8-one by decarboxylative cyclization reaction with isatoic anhydride using tetrabutylammonium iodide (TBAI).
- 1-naphtholyl tetrahydroisoquinoline by aza-Friedel-Crafts reaction with various naphthols.
- 3,4-dihydroisoquinoline pseudo bases, which are employed as starting materials for the preperation of 3-benzazepine derivatives.
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 2 Dermal - Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2
Storage Class Code
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
Electrosynthesis of polycyclic quinazolinones and rutaecarpine from isatoic anhydrides and cyclic amines
Chen Xingyu, et al.
Royal Society of Chemistry Advances, 10(72), 44382-44386 (2020)
A facile, one pot method for the synthesis of 4-acyl-1, 2-dihydro-3-benzazepines, based on the ring expansion of natural and synthetic 3, 4-dihydroisoquinoline pseudo bases
Kartsev VG, et al.
Tetrahedron Letters, 56(50), 6988-6993 (2015)
Solvent-free direct aza-Friedel-Crafts reactions between 3, 4-dihydroisoquinoline and 1-or 2-naphthols
MacLeod PD, et al.
Tetrahedron Letters, 47(38), 6791-6794 (2006)
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.





![Benzo[h]quinoline 97%](/deepweb/assets/sigmaaldrich/product/structures/344/715/928932d2-4ca4-4402-b56c-85a80100ce17/640/928932d2-4ca4-4402-b56c-85a80100ce17.png)
