모든 사진(2)
About This Item
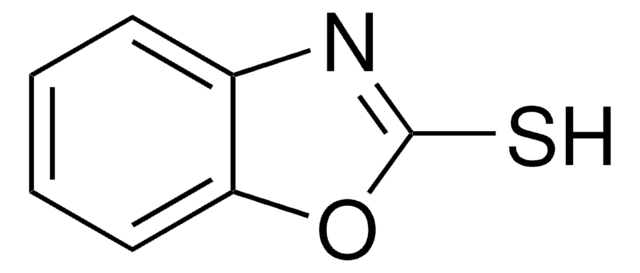
실험식(Hill 표기법):
C7H5N4NaS
CAS Number:
Molecular Weight:
200.20
EC Number:
MDL number:
UNSPSC 코드:
12352100
PubChem Substance ID:
NACRES:
NA.22
추천 제품
분석
98%
mp
>300 °C (lit.)
solubility
water: soluble 1g/15ml, clear
SMILES string
[Na]Sc1nnnn1-c2ccccc2
InChI
1S/C7H6N4S.Na/c12-7-8-9-10-11(7)6-4-2-1-3-5-6;/h1-5H,(H,8,10,12);/q;+1/p-1
InChI key
RSZMKAPXKXEWBY-UHFFFAOYSA-M
일반 설명
1-Phenyl-1H-tetrazole-5-thiol sodium salt is a tetrazole derivative. The kinetics of its reaction with Diel′s-Alder adducts of 1,4-benzoquinone has been investigated.
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
Kinetics of the reaction of 1-phenyl-1H-tetrazole-5-thiol sodium salt with less substituted 1, 4-naphthoquinone 2, 3-epoxides.
O'Brien DF.
The Journal of Organic Chemistry, 33(1), 262-265 (1968)
Mohammad M Khodaei et al.
The Journal of organic chemistry, 73(7), 2527-2532 (2008-03-05)
In the presence of 1-phenyl-5-mercaptotetrazole as a nucleophile, electrochemical oxidations of 1,2- and 1,4-dihydroxybenzenes have been investigated in aqueous solution using cyclic voltammetry and controlled-potential coulometry. The voltammetric results indicate that an electrooxidative/Michael-type sequential reaction occurs between the mercaptide anion
J Kejha et al.
Ceskoslovenska farmacie, 39(7), 294-298 (1990-09-01)
For the investigation of new anti-inflammatory drugs, 1-phenyl-5-mercaptotetrazole (I) was selected as the principal structure. The purpose itself lay in an alkylation of the mercapto group with different remainders. In some cases at the same time a substituent was introduced
K Nesmĕrák et al.
Folia microbiologica, 45(2), 138-142 (2001-03-29)
The antifungal effect of substituted 1-phenyl-5-mercaptotetrazoles was tested with Candida tropicalis, C. pseudotropicalis, C. mogii, Trichosporon cutaneum, Cryptococcus albidus and S. cerevisiae. Candida strains exhibited the lowest sensitivity to the compounds; the most sensitive was S. cerevisiae. The MIC values
[Information from the Soviet Toxicology Center].
Gigiena truda i professional'nye zabolevaniia, (10)(10), 55-56 (1980-10-01)
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.