추천 제품
분석
98%
형태
solid
mp
128-131 °C (lit.)
SMILES string
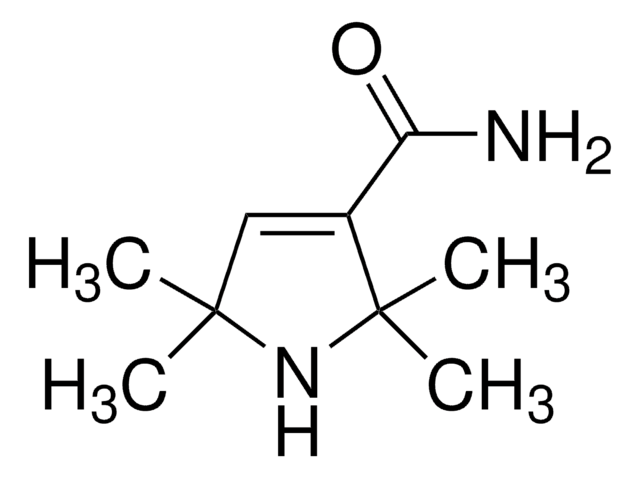
CC1(C)CC(C(N)=O)C(C)(C)N1
InChI
1S/C9H18N2O/c1-8(2)5-6(7(10)12)9(3,4)11-8/h6,11H,5H2,1-4H3,(H2,10,12)
InChI key
POAGFQOGFRYOFM-UHFFFAOYSA-N
일반 설명
2,2,5,5-Tetramethyl-3-pyrrolidinecarboxamide (2,2,5,5-Tetramethylpyrrolidine-3-carboxamide) is a carboxamide of hydrogenated pyrrole derivative. Its synthesis by the hydrogenation of 2,2,5,5-tetramethyl-3-pyrroline-3-carboxamide has been reported. The antiarrhythmic activity of some of the derivatives of 2,2,5,5-tetramethylpyrrolidine-3-carboxamide has been evaluated.
2,2,5,5-Tetramethyl-3-pyrrolidinecarboxamide, a pyrrolidine derivative, is a cyclic (five-membered ring) secondary amine having four carbon atoms and one nitrogen atom. Various physical properties (freezing point, boiling point, density and refractive index) of 2,2,5,5-tetramethyl-3-pyrrolidinecarboxamidehave been reported.
애플리케이션
2,2,5,5-Tetramethyl-3-pyrrolidinecarboxamide may be used in the synthesis of its nitroxide, 3-carbamoyl-2,2,5,5-tetramethyl-1-pyrrolidinyloxy free radical.
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
Nitroxide free radicals in the hydrogenated pyrrole series.
Rozantsev EG, et al.
Russian Chemical Bulletin, 15(4), 638-641 (1966)
Yaws CL.
The Yaws Handbook of Physical Properties for Hydrocarbons and Chemicals, 260-260 (2015)
O H Hankovszky et al.
Journal of medicinal chemistry, 29(7), 1138-1152 (1986-07-01)
N-(omega-Aminoalkyl)-2,2,5,5-tetramethyl-3-pyrroline- or -pyrrolidine-3-carboxamides were acylated on the primary amino group of the side chain by means of reactive acid derivatives (acid chlorides, activated esters, phthalic anhydrides, phthalimide, 2-alkyl-4H-3,1-benzoxazin-4-ones) or they were alkylated by forming the Schiff bases and subsequent sodium
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.