추천 제품
분석
99%
형태
solid
광학 활성
[α]20/D −145°, c = 2 in acetone
광학 순도
ee: 99% (HPLC)
mp
75-77 °C (lit.)
SMILES string
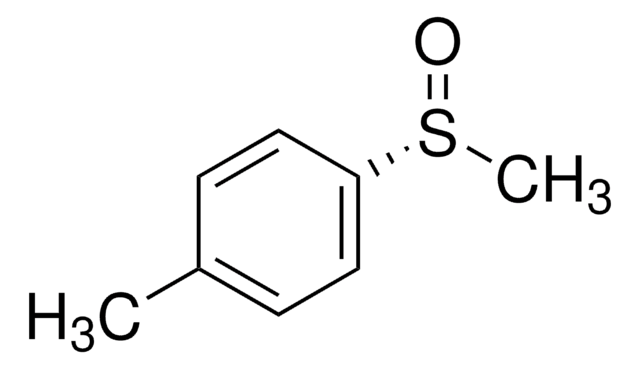
Cc1ccc(cc1)S(C)=O
InChI
1S/C8H10OS/c1-7-3-5-8(6-4-7)10(2)9/h3-6H,1-2H3/t10-/m0/s1
InChI key
FEVALTJSQBFLEU-JTQLQIEISA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
애플리케이션
(S)-(-)-Methyl p-tolyl sulfoxide can be used as a nucleophilic reagent to synthesize:
- Optically active β-disulfoxides by reacting with arenesulfinic esters via formation of α-sulfinylcarbanion.
- α-substituted N-hydroxylamines by treating with nitrones via preparation of (S)-(-)-methyl p-tolyl sulfoxide anion.
- 2-O-benzyl-3,4-O-isopropylidene-L-erythrose by one-carbon homologation of 2,3-O-isopropylidene-L-glyceraldehyde.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type N95 (US)
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
β -Disulfoxides. II. The Preparation of Some Optically Active β -Disulfoxides
Kunieda N, et al.
Bulletin of the Chemical Society of Japan, 49(1), 256-259 (1976)
A highly stereoselective synthesis of d-erythrose derivatives by one-carbon homologation of 2, 3-O-isopropylidene-d-glyceraldehyde with (R)-methyl p-tolyl sulfoxide.
Arroyo-Gomez Y, et al.
Tetrahedron Asymmetry, 11(3), 789-796 (2000)
The reaction of nitrones with (R)-(+)-methyl p-tolyl sulfoxide anion; asymmetric synthesis of optically active secondary amines.
Murahashi S-I, et al.
Tetrahedron Letters, 34(16), 2645-2648 (1993)
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.