추천 제품
분석
98%
형태
solid
mp
123-126 °C (lit.)
solubility
95% ethanol: soluble 50 mg/mL, clear, light yellow
SMILES string
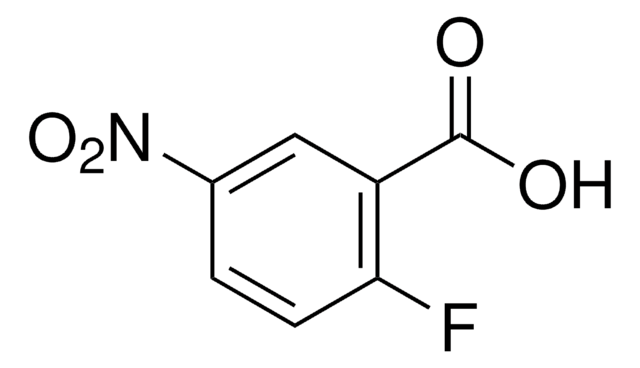
OC(=O)c1ccc(F)c(c1)[N+]([O-])=O
InChI
1S/C7H4FNO4/c8-5-2-1-4(7(10)11)3-6(5)9(12)13/h1-3H,(H,10,11)
InChI key
BOJWTAQWPVBIPG-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
애플리케이션
4-Fluoro-3-nitrobenzoic acid was used:
- as starting reagent in the preparation of novel benzimidazoles having antimycobacterial activity
- in preparation of series of novel acetylcholinesterase (AChE) and butyrylcholinesterase (BChE) inhibitors containing benzimidazole core structure
- in preparation of bis(heterocyclic) skeletal precursors for the Pictet-Spengler reaction
- in solid-phase synthesis of trisubstituted [1,3,5]triazino[1,2-a]benzimidazole-2,4(3H,10H)-diones
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
Yeong Keng Yoon et al.
European journal of medicinal chemistry, 93, 614-624 (2014-07-06)
A total of 51 novel benzimidazoles were synthesized by a 4-step reaction starting from basic compound 4-fluoro-3-nitrobenzoic acid under relatively mild reaction conditions. The structure of the novel benzimidazoles was confirmed by mass spectra as well as (1)H NMR spectroscopic
Yeong Keng Yoon et al.
Bioorganic chemistry, 49, 33-39 (2013-07-28)
Two series of novel acetylcholinesterase (AChE) and butyrylcholinesterase (BChE) inhibitors containing benzimidazole core structure were synthesized by a four-step reaction pathway starting from 4-fluoro-3-nitrobenzoic acid as the basic compound. The structure of the novel benzimidazoles was characterized and confirmed by
J C Wijkmans et al.
Molecular diversity, 3(2), 117-120 (1997-01-01)
4-Fluoro-3-nitrobenzoic acid attached to a solid support was shown to react under mild conditions with a wide range of functionalized phenols to yield, after cleavage, the corresponding biaryl ethers in excellent purity. In a similar fashion, biaryl thioethers could be
Gérard Klein et al.
Journal of combinatorial chemistry, 4(4), 345-351 (2002-07-09)
An efficient method for the solid-phase synthesis of trisubstituted [1,3,5]triazino[1,2-a]benzimidazole-2,4(3H,10H)-diones from resin-bound amino acids is described. N-acylation of the primary amine of a resin-bound amino acid with 4-fluoro-3-nitrobenzoic acid, followed by displacement of the fluoro group and reduction of the
Chih-Hau Chen et al.
Chemistry, an Asian journal, 6(6), 1557-1565 (2011-04-08)
A novel strategy for an unconventional Pictet-Spengler reaction has been developed for the regioselective cyclization of the imidazole ring system at the C2 position. The developed strategy was utilized to develop a diversity-oriented parallel synthesis for bis(heterocyclic) skeletal novel analogs
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.