모든 사진(1)
About This Item
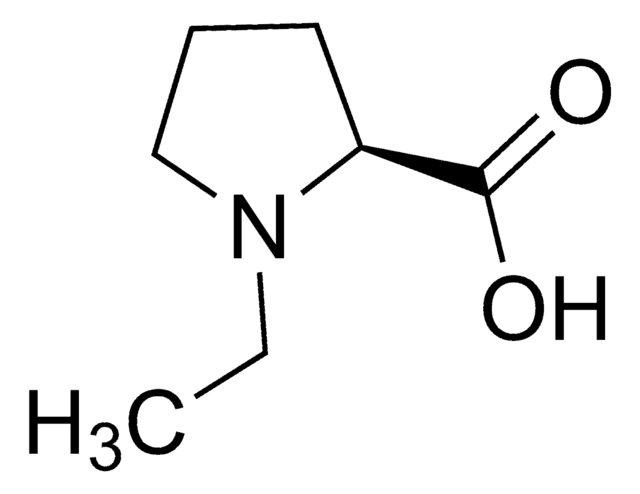
실험식(Hill 표기법):
C6H11NO2
CAS Number:
Molecular Weight:
129.16
Beilstein:
4350211
MDL number:
UNSPSC 코드:
12352209
PubChem Substance ID:
NACRES:
NA.22
추천 제품
분석
≥98.0% (TLC)
반응 적합성
reaction type: solution phase peptide synthesis
응용 분야
peptide synthesis
SMILES string
C[C@]1(CCCN1)C(O)=O
InChI
1S/C6H11NO2/c1-6(5(8)9)3-2-4-7-6/h7H,2-4H2,1H3,(H,8,9)/t6-/m0/s1
InChI key
LWHHAVWYGIBIEU-LURJTMIESA-N
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type N95 (US)
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
J H Welsh et al.
FEBS letters, 297(3), 216-220 (1992-02-10)
The ability of (S)-alpha-methylproline (alpha-MePro) to stabilise reverse-turn conformations in the peptide hormone bradykinin (BK = Arg1-Pro2-Pro3-Gly4-Phe5-Ser6-Pro7-Phe8-Arg9) has been investigated. Two BK analogues containing alpha-MePro at position 3 or position 7 were synthesised and their conformations in aqueous solution investigated
G Lubec et al.
Life sciences, 57(24), 2245-2252 (1995-01-01)
Collagen type I is the major protein of bone matrix and significantly reduced in osteoporosis. We tested the effect of alpha - methyl - proline on collagen synthesis in the model of the ovariectomized rat. Collagen synthesis was studied at
S Thaisrivongs et al.
Journal of medicinal chemistry, 30(3), 536-541 (1987-03-01)
A structure-activity analysis of peptides containing backbone C alpha-methyl modification at the P4 site of the angiotensinogen sequence led to the discovery of potent renin inhibitors with apparent in vitro metabolic stability. Boc-alpha-MePro-Phe-His-Leu psi[CHOHCH2]Val-Ile-Amp dicitrate (Va) is a potent inhibitor
Alessandro Moretto et al.
Biopolymers, 89(5), 465-470 (2007-09-07)
Methylation at the C(alpha)-position of a Pro residue was expected to lock the preceding tertiary amide (omega) torsion angle of the resulting (alphaMe)Pro to the trans disposition and to restrict the phi,psi surface to the single region where the 3(10)/alpha-helices
Matteo De Poli et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 15(32), 8015-8025 (2009-07-07)
C(alpha)-methyl-L-proline, or L-(alphaMe)Pro, is probably the most conformationally constrained alpha-amino acid. In particular, its omega and phi torsion angles are restricted to about 180 and -60 degrees, respectively, and only three ranges of values are theoretically available for psi in
문서
Proline analogues are promising candidates for tuning the biological, pharmaceutical, or physicochemical properties of naturally occuring, as well as de novo designed, linear, and, cyclic peptides.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.