추천 제품
규정 준수
suitable for FDA C-010.02
Quality Level
분석
97%
양식
liquid
density
0.92 g/mL at 25 °C (lit.)
작용기
ketone
저장 온도
2-8°C
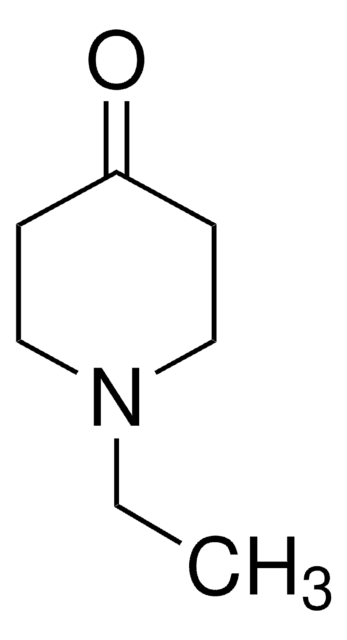
SMILES string
CN1CCC(=O)CC1
InChI
1S/C6H11NO/c1-7-4-2-6(8)3-5-7/h2-5H2,1H3
InChI key
HUUPVABNAQUEJW-UHFFFAOYSA-N
애플리케이션
N-Methyl-4-piperidone can be used as a reactant to prepare:
- Spiropiperidine rings by reacting with malononitrile and electrophiles or Michael acceptors.
- (3E,5E)-1-Methyl-3,5-bis(phenylmethylene)-4-piperidinone by reacting with benzaldehyde via Michael addition, followed by intramolecular O-cyclization/elimination sequential reactions.
- N,N′-Dimethylbispidinone by utilizing a double Mannich condensation method.
이미 열람한 고객
Analogs of sparteine. I. Reexamination of the reaction of N-methyl-4-piperidone with formaldehyde and methylamine. Revised synthesis of N, N'-dimethylbispidinone
Smissman EE, et al.
The Journal of Organic Chemistry, 40, 251-252 (1975)
Novel route to spiropiperidines using N-methyl-4-piperidone, malononitrile and electrophiles
Lakshmi NV, et al.
Tetrahedron Letters, 53, 1282-1286 (2012)
Bin-Rong Yao et al.
European journal of medicinal chemistry, 167, 187-199 (2019-02-17)
To get new anti-hepatoma agents with anti-inflammatory activity and hypotoxicity, a series of dissymmetric pyridyl-substituted 3,5-bis(arylidene)-4-piperidones (BAPs, 25-82) were designed and synthesized. Many of them exhibited potential anti-hepatoma properties against human hepatocellular carcinoma cell lines (HepG2, QGY-7703, SMMC-7721) and hypotoxicity
A facile tandem Michael addition/O-cyclization/elimination route to novel chromeno [3, 2-c] pyridines
Sumesh RV, et al.
Molecular Diversity, 19, 233-249 (2015)
Analogs of sparteine. I. A reexamination of the reaction of n-methyl-4-piperidone with formaldehyde and methylamine. A revised synthesis of n,n'-dimethylbispidinone.
E E Smissman et al.
The Journal of organic chemistry, 40(2), 251-252 (1975-01-24)
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.