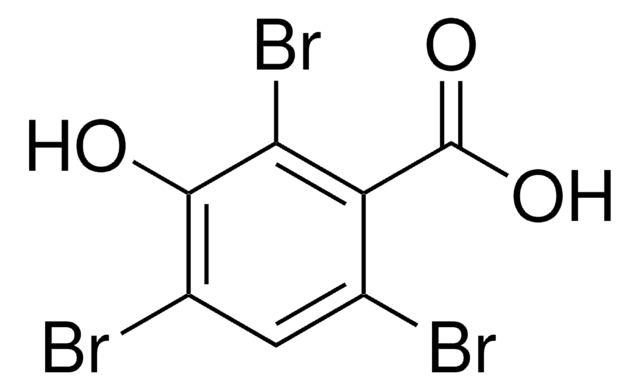
11460
8-Azaxanthine monohydrate
≥98.0% (HPLC)
Synonym(s):
2,6-Dihydroxy-8-azapurine
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C4H3N5O2 · H2O
CAS Number:
Molecular Weight:
171.11
Beilstein:
10424
EC Number:
MDL number:
UNSPSC Code:
41106305
PubChem Substance ID:
NACRES:
NA.25
Recommended Products
Assay
≥98.0% (HPLC)
form
solid
SMILES string
O.O=C1NC(=O)c2[nH]nnc2N1
InChI
1S/C4H3N5O2.H2O/c10-3-1-2(8-9-7-1)5-4(11)6-3;/h(H3,5,6,7,8,9,10,11);1H2
InChI key
VKEGPGRANAWNIN-UHFFFAOYSA-N
Application
8-Azaxanthine monohydrate has been used to determine the crystal and molecular structure of 1,3-dimethyl-8-azaxanthine (HDAX) monohydrate by X-ray diffraction.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Laurent Fraisse et al.
Analytical biochemistry, 309(2), 173-179 (2002-11-05)
Urate oxidase (E.C.1.7.3.3; uricase, urate oxygen oxidoreductase) is an enzyme of the purine breakdown pathway that catalyzes the oxidation of uric acid in the presence of oxygen to allantoin and hydrogen peroxide. A 96-well plate assay measurement of urate oxidase
Molecular orbital study of 8-azaxanthine derivatives and crystal structure of 1,3-dimethyl-8-azaxanthine monohydrate
Purificacion Sanchez, M., et al.
Journal of Molecular Structure, 344, 257-264 (1995)
5-Methylsulfanyl-3H-1,2,3-triazolo[4,5-d]pyrimidin-7(6H)-one (2-methylthio-8-azaxanthine) monohydrate
Maldonado, C.R., et al.
Acta Crystallographica Section C, Crystal Structure Communications, 62, 489-491 (2006)
P Franchetti et al.
Journal of medicinal chemistry, 37(18), 2970-2975 (1994-09-02)
A series of 1,3-dimethyl- and 1,3-dipropyl-8-azaxanthines, substituted at the N8 or N7 position with substituents which usually increase the affinity of the xanthines for the adenosine receptors, was synthesized and studied in radioligand binding experiments. The substitution of CH with
J Giełdanowski
Archivum immunologiae et therapiae experimentalis, 28(3), 469-474 (1980-01-01)
The influence of antibiotics and purine and pyrimidine analogues on cells forming rosettes EA and EAC was evaluated in vitro. At the same time pH of solutions of examined immunosuppressors was estimated and a series of experiments with a buffer
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service