All Photos(2)
About This Item
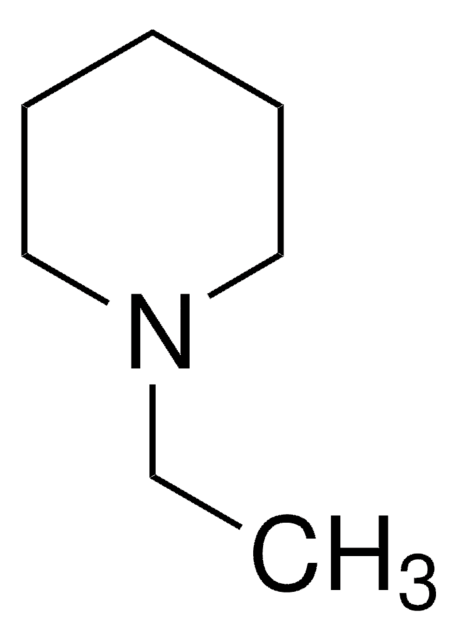
Empirical Formula (Hill Notation):
C8H17N
CAS Number:
Molecular Weight:
127.23
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
form
liquid
refractive index
n20/D 1.44 (lit.)
bp
155-157 °C/754 mmHg (lit.)
density
0.814 g/mL at 25 °C (lit.)
SMILES string
CCCCN1CCCC1
InChI
1S/C8H17N/c1-2-3-6-9-7-4-5-8-9/h2-8H2,1H3
InChI key
JSHASCFKOSDFHY-UHFFFAOYSA-N
Related Categories
Application
1-Butylpyrrolidine has been used in:
- microwave-assisted synthesis of ionic liquid precursor, 1-butyl-1-methylpyrrolidinium methylcarbonate
- [N,N-methylbutylpyrrolidinium] thiosalicylate, ionic liquid
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 3 Oral - Flam. Liq. 3
Storage Class Code
3 - Flammable liquids
WGK
WGK 2
Flash Point(F)
96.8 °F - closed cup
Flash Point(C)
36 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Giridhar Pulletikurthi et al.
Dalton transactions (Cambridge, England : 2003), 46(2), 455-464 (2016-12-14)
The mixtures of 1-butylpyrrolidine and ZnCl
Synthesis, characterization and thermal properties of thiosalicylate ionic liquids.
Wilfred CD and Mustafa FB.
Journal of Chemical Sciences (Bangalore), 125(6), 1511-1515 (2013)
Giridhar Pulletikurthi et al.
Chemistry, an Asian journal, 12(20), 2684-2693 (2017-08-05)
Electrostatic interactions are characteristic of ionic liquids (ILs) and play a pivotal role in determining the formation of species when solutes are dissolved in them. The formation of new species/complexes has been investigated for certain ILs. However, such investigations have
Optimised microwave-assisted synthesis of methylcarbonate salts: a convenient methodology to prepare intermediates for ionic liquid libraries.
Holbrey JD, et al.
Green Chemistry, 12(3), 407-413 (2010)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service