All Photos(1)
About This Item
Empirical Formula (Hill Notation):
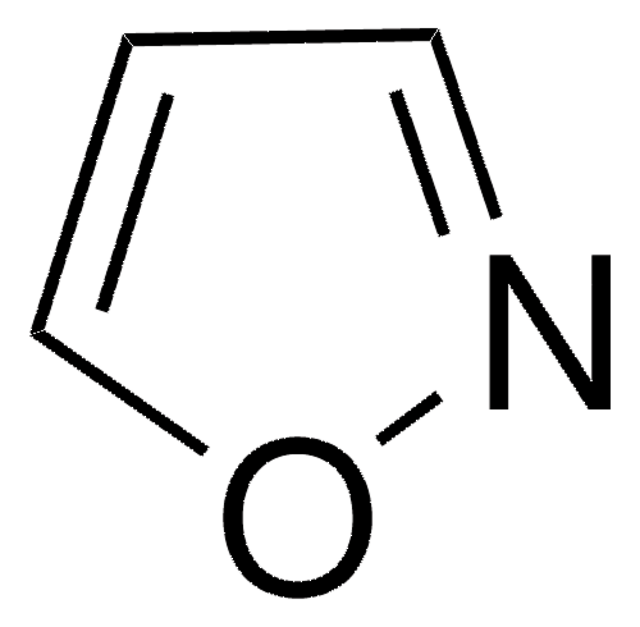
C3H3NO
CAS Number:
Molecular Weight:
69.06
Beilstein:
103851
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
refractive index
n20/D 1.425 (lit.)
bp
69-70 °C (lit.)
mp
−87-−84 °C (lit.)
density
1.05 g/mL at 25 °C (lit.)
SMILES string
c1cocn1
InChI
1S/C3H3NO/c1-2-5-3-4-1/h1-3H
InChI key
ZCQWOFVYLHDMMC-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Oxazole is the parent molecule for a large class of heterocyclic aromatic compounds. It is a weak base that can be used as an electron-deficient diene in the Diels-Alder cycloaddition reaction. It undergoes nitration, sulfonation, halogenation, Friedel-Crafts alkylation, and acylation.
accessory
Product No.
Description
Pricing
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Dam. 1 - Flam. Liq. 2
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
66.2 °F - closed cup
Flash Point(C)
19 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Reem A Masri et al.
Insects, 12(2) (2021-02-11)
Long-read sequencing technologies have opened up new avenues of research on the mosquito genome biology, enabling scientists to better understand the remarkable abilities of vectors for transmitting pathogens. Although new genome mapping technologies such as Hi-C scaffolding and optical mapping
Ilya Lyagin et al.
Molecules (Basel, Switzerland), 24(13) (2019-06-30)
Mycotoxins are highly dangerous natural compounds produced by various fungi. Enzymatic transformation seems to be the most promising method for detoxification of mycotoxins. This review summarizes current information on enzymes of different classes to convert various mycotoxins. An in-depth analysis
Lori M Culberson et al.
Physical chemistry chemical physics : PCCP, 16(9), 3964-3972 (2014-01-22)
Bond breaking is a challenging problem in both experimental and theoretical chemistry, due to the transient nature and multi-configurational electronic structure of dissociating molecules. We use anion photodetachment to probe the diradical interactions in the ring-opening reaction of oxazole and
Fengjiao Zhang et al.
Toxicological sciences : an official journal of the Society of Toxicology, 140(1), 118-134 (2014-04-23)
The generation of reactive oxygen species (ROS) has been implicated in the pathogenesis of renal ischemia/reperfusion injury, and many other pathological conditions. DNA strand breaks caused by ROS lead to the activation of poly(ADP-ribose)polymerase-1 (PARP-1), the excessive activation of which
Systematic scientific study of 1, 3-oxazole derivatives as a useful lead for pharmaceuticals: A review
Joshi S, et al.
The pharma innovation journal, 6, 109-109 (2017)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service