All Photos(1)
About This Item
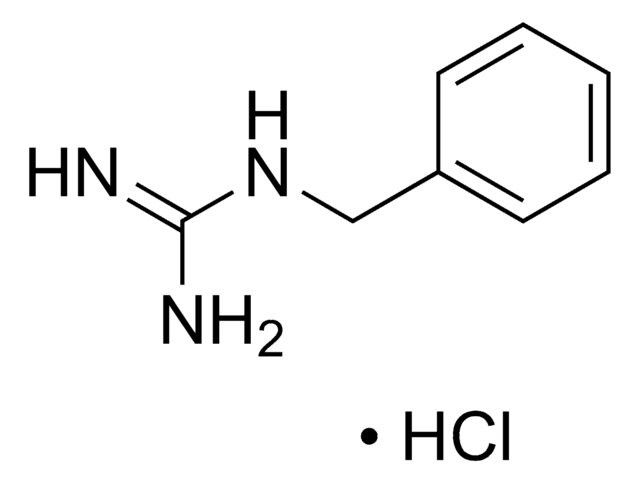
Linear Formula:
CH3NHC(=NH)NH2·HCl
CAS Number:
Molecular Weight:
109.56
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
form
solid
solubility
water: soluble 50 mg/mL, clear
functional group
amine
SMILES string
Cl.CNC(N)=N
InChI
1S/C2H7N3.ClH/c1-5-2(3)4;/h1H3,(H4,3,4,5);1H
InChI key
VJQCNCOGZPSOQZ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
Methylguanidine hydrochloride can be used to prepare:
- Biaryl derivatives as BACE1 inhibitors.
- Modified xylose, which is used in the synthesis of biodegradable composite hydrogels.
- Methylguanidinium borohydride ionic liquid, which is applicable as a hydrogen storage material.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
A non-covalent strategy for montmorillonite/xylose self-healing hydrogels
Qi X, et al.
Royal Society of Chemistry Advances, 5(51), 41006-41012 (2015)
Jared N Cumming et al.
Bioorganic & medicinal chemistry letters, 22(7), 2444-2449 (2012-03-07)
From an initial lead 1, a structure-based design approach led to identification of a novel, high-affinity iminohydantoin BACE1 inhibitor that lowers CNS-derived Aβ following oral administration to rats. Herein we report SAR development in the S3 and F' subsites of
Methylguanidinium borohydride: an ionic-liquid-based hydrogen-storage material
Doroodian A, et al.
Angewandte Chemie (International ed. in English), 49(10), 1871-1873 (2010)
Jovana Vušurović et al.
ChemistryOpen, 6(6), 739-750 (2017-12-12)
Interactions of ribonucleic acid (RNA) with guanidine and guanidine derivatives are important features in RNA-protein and RNA-drug binding. Here we have investigated noncovalently bound complexes of an 8-nucleotide RNA and six different ligands, all of which have a guanidinium moiety
Structure based design of iminohydantoin BACE1 inhibitors: identification of an orally available, centrally active BACE1 inhibitor
Cumming JN, et al.
Bioorganic & Medicinal Chemistry Letters, 22(7), 2444-2449 (2012)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service