おすすめの製品
アッセイ
≥98%
形状
powder
色
white to beige
溶解性
DMSO: 2 mg/mL, clear
保管温度
−20°C
SMILES記法
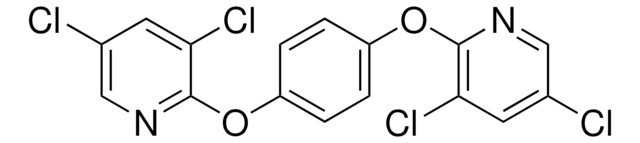
O=C(CN(CC)CC)N1C2=C(C=CC=C2)CCC3=C1C=C(NC(OCC)=O)C=C3
生物化学的/生理学的作用
CINPA1 is a ligand-binding domain (LBD)-targeting constitutive androstane receptor (CAR) inhibitor that prevents CAR recruitment to promoter regions of regulated genes (by >85%; 1 μM CINPA1 against 0.1 μM CITCO-induced CAR recruitment to CYP2B6 or CYP3A4 promoters; human hepatocytes) by altering CAR-coregulator interactions. CINPA1 inhibits CAR-mediated transcription in a potent (IC50 = 70 nM; HepG2-hCAR1 CYP2B6-luc reporter cells) and selective manner with only weak PXR antagonist potency (68% inhibition by 18 μM CINPA1 against 5 μM rifampicin; HepG2-PXR reporter cells) and no cytotoxicity or agonist/antagonist activity toward FXR, GR, LXRα/β, PPARγ, RXRα/β, VDR.
保管分類コード
11 - Combustible Solids
WGK
WGK 3
引火点(°F)
Not applicable
引火点(℃)
Not applicable
適用法令
試験研究用途を考慮した関連法令を主に挙げております。化学物質以外については、一部の情報のみ提供しています。 製品を安全かつ合法的に使用することは、使用者の義務です。最新情報により修正される場合があります。WEBの反映には時間を要することがあるため、適宜SDSをご参照ください。
Jan Code
SML2468-BULK:
SML2468-VAR:
SML2468-25MG:
SML2468-5MG:
試験成績書(COA)
製品のロット番号・バッチ番号を入力して、試験成績書(COA) を検索できます。ロット番号・バッチ番号は、製品ラベルに「Lot」または「Batch」に続いて記載されています。
Wenwei Lin et al.
European journal of medicinal chemistry, 108, 505-528 (2015-12-31)
Constitutive androstane receptor (CAR, NR1I3) and pregnane X receptor (PXR, NR1I2) are master regulators of endobiotic and xenobiotic metabolism and disposition. Because CAR is constitutively active in certain cellular contexts, inhibiting CAR might reduce drug-induced hepatotoxicity and resensitize drug-resistant cancer
Milu T Cherian et al.
Biochemical pharmacology, 152, 211-223 (2018-04-03)
The constitutive androstane receptor (CAR) and pregnane X receptor (PXR) are xenobiotic sensors that regulate the expression of drug-metabolizing enzymes and efflux transporters. CAR activation promotes drug elimination, thereby reducing therapeutic effectiveness, or causes adverse drug effects via toxic metabolites.
Baddreddine Boussadia et al.
Experimental neurology, 283(Pt A), 39-48 (2016-06-01)
Nuclear receptors (NRs) are a group of transcription factors emerging as players in normal and pathological CNS development. Clinically, an association between the constitutive androstane NR (CAR) and cognitive impairment was proposed, however never experimentally investigated. We wished to test
Milu T Cherian et al.
Drug metabolism and disposition: the biological fate of chemicals, 44(11), 1759-1770 (2016-08-16)
The constitutive androstane receptor (CAR) regulates the expression of genes involved in drug metabolism and other processes. A specific inhibitor of CAR is critical for modulating constitutive CAR activity. We recently described a specific small-molecule inhibitor of CAR, CINPA1 (ethyl
Bryan Mackowiak et al.
Molecular pharmacology, 92(1), 75-87 (2017-04-27)
The constitutive androstane receptor (CAR) plays an important role in xenobiotic metabolism, energy homeostasis, and cell proliferation. Antagonism of the CAR represents a key strategy for studying its function and may have potential clinical applications. However, specific human CAR (hCAR)
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)