SAE0093
Beta-1,4-galactosyltransferase 1
B4GALT1 human recombinant, expressed in HEK 293 cells, 2000 units/mg protein
Synonym(s):
Beta-1,4-GalTase 1, Beta4Gal-T1, UDP-Gal:beta-GlcNAc beta-1,4-galactosyltransferase 1, UDP-galactose:beta-N-acetylglucosamine beta-1,4-galactosyltransferase 1, b4Gal-T1
About This Item
Recommended Products
recombinant
expressed in HEK 293 cells
Assay
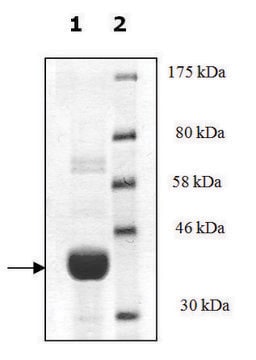
95% (SDS-PAGE)
form
lyophilized powder
specific activity
2000 units/mg protein
shipped in
ambient
storage temp.
−20°C
General description
Application
Biochem/physiol Actions
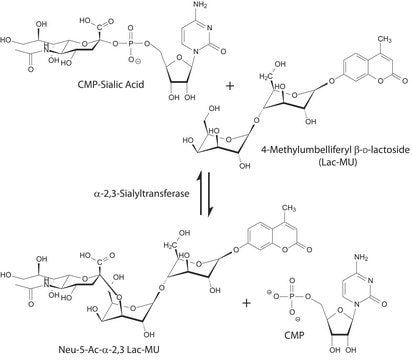
A major function of B4GALT1 is the addition of β(1→4) linked galactose residues to oligosaccharide acceptors with terminal N-acetylglucosamine residues. This is a late elongation step in the N-glycan processing pathway.B4GALT1 enzymatic activity is widely distributed in the vertebrate kingdom, in both mammals and non-mammals, including avians and amphibians.B4GALT1 enzymatic activity has also been demonstrated in a subset of plants which diverged from animals an estimated 1 billion years ago.B4GALT1 interacts with α-lactalbumin (LA), a protein expressed in the mammary gland during lactation, to form the lactose synthase (LS) complex that transfers galactose from UDP-α-D-Gal to glucose, producing the lactose secreted in milk.Defects in B4GALT1 are the cause of congenital disorder of glycosylation type 2D (CDG2D).Glomerular B4GALT1 expression has been found to be increased in IgA nephropathy. IgA binding and IgA-induced mesangial cell phosphorylation of spleen tyrosine kinase and IL-6 synthesis were inhibited by a panel of β(1→4) galactosyltransferase-specific antibodies, which suggests that IgA binds to the catalytic domain of β(1→4) galactosyltransferase.
Unit Definition
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Regulatory Listings
Regulatory Listings are mainly provided for chemical products. Only limited information can be provided here for non-chemical products. No entry means none of the components are listed. It is the user’s obligation to ensure the safe and legal use of the product.
JAN Code
SAE0093-50UG:
SAE0093-VAR:
SAE0093-BULK:
SAE0093-50UG-PW:
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Explore tools for glycosyltransferase synthesis and modification of glycans, such as glycosyltransferases and nucleotide sugar donors.
Enzymatic glycosyltransferase specificity challenges the one enzyme-one linkage concept.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service