416916
Dicyclohexyliodoborane
97%
Synonym(s):
Dicyclohexylboryl iodide
About This Item
Recommended Products
Assay
97%
form
liquid
reaction suitability
reagent type: reductant
bp
198-200 °C/1.25 mmHg (lit.)
density
1.325 g/mL at 25 °C (lit.)
SMILES string
IB(C1CCCCC1)C2CCCCC2
InChI
1S/C12H22BI/c14-13(11-7-3-1-4-8-11)12-9-5-2-6-10-12/h11-12H,1-10H2
InChI key
RWFGGTOYIFQXAO-UHFFFAOYSA-N
Application
- For the enolboration of esters and tertiary amides to synthesize corresponding Z or E enolates.[1][2]
- In the stereoselective preparation of β-hydroxy-α-trifluoromethyl carboxylic acids via haloborane-mediated diastereoselective aldol addition of aldehydes with trifluoropropanoic acid.[3]
- In the total synthesis of pordamacrine A,[4] and trocheliophorolide D.[5]
- Preparation of vinyloxyboranes
Regulatory Listings
Regulatory Listings are mainly provided for chemical products. Only limited information can be provided here for non-chemical products. No entry means none of the components are listed. It is the user’s obligation to ensure the safe and legal use of the product.
FSL
Group 4: Flammable liquids
Type 2 petroleums
Hazardous rank III
Water insoluble liquid
ISHL Indicated Name
Substances Subject to be Indicated Names
ISHL Notified Names
Substances Subject to be Notified Names
JAN Code
416916-25ML:4548173993959
416916-VAR:
416916-BULK:
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Active Filters
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service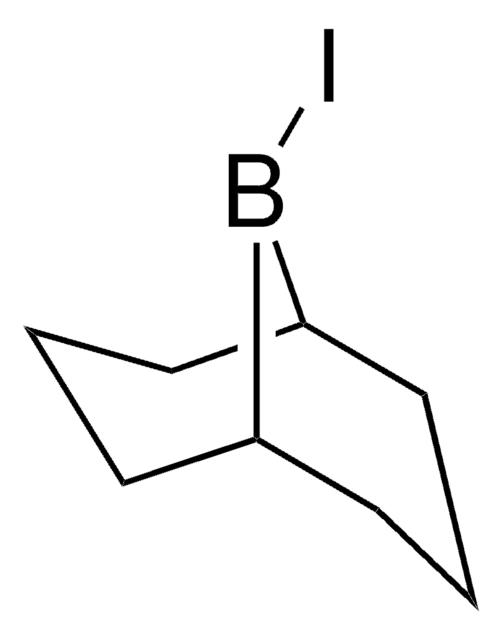
![9-Borabicyclo[3.3.1]nonane solution 0.5 M in THF](/deepweb/assets/sigmaaldrich/product/structures/180/891/8b64e597-269d-4780-98b6-40889dfd06b9/640/8b64e597-269d-4780-98b6-40889dfd06b9.png)



![9-Borabicyclo[3.3.1]nonane dimer](/deepweb/assets/sigmaaldrich/product/structures/203/431/624973a6-aec1-4b23-b6c4-013285ac418c/640/624973a6-aec1-4b23-b6c4-013285ac418c.png)




