All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C7H6N2
CAS Number:
Molecular Weight:
118.14
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
99%
form
liquid
refractive index
n20/D 1.626 (lit.)
bp
103 °C/1 mmHg (lit.)
density
1.165 g/mL at 25 °C (lit.)
SMILES string
c1ccn2ccnc2c1
InChI
1S/C7H6N2/c1-2-5-9-6-4-8-7(9)3-1/h1-6H
InChI key
UTCSSFWDNNEEBH-UHFFFAOYSA-N
Related Categories
General description
In vivo anti-trypanosomal activity of imidazo[1,2-a]pyridiness in the STIB900 mouse model has been investigated.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
235.4 °F - closed cup
Flash Point(C)
113 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Martina Hieke et al.
Bioorganic & medicinal chemistry letters, 22(5), 1969-1975 (2012-02-14)
A novel class of 5-lipoxygenase (5-LO) inhibitors characterized by a central imidazo[1,2-a]pyridine scaffold, a cyclohexyl moiety and an aromatic system, is presented. This scaffold was identified in a virtual screening study and exhibits promising inhibitory potential on the 5-LO. Here
Kyle A Emmitte et al.
Bioorganic & medicinal chemistry letters, 19(3), 1004-1008 (2008-12-23)
The optimization of imidazo[1,2-a]pyridine inhibitors as potent and selective inhibitors of IGF-1R is presented. Further optimization of oral exposure in mice is also discussed. Detailed selectivity, in vitro activity, and in vivo PK profiles of an optimized compound is also
Sophie Marhadour et al.
European journal of medicinal chemistry, 58, 543-556 (2012-11-21)
A novel series of 2,3-diarylimidazo[1,2-a]pyridines was synthesized and evaluated for their antileishmanial activities. Four derivatives exhibited good activity against the promastigote and intracellular amastigote stages of Leishmania major, coupled with a low cytotoxicity against the HeLa human cell line. The
Joanna M Wisniewska et al.
Biochemical pharmacology, 83(2), 228-240 (2011-10-27)
5-Lipoxygenase (5-LO) is a crucial enzyme of the arachidonic acid (AA) cascade and catalyzes the formation of bioactive leukotrienes (LTs) which are involved in inflammatory diseases and allergic reactions. The pathophysiological effects of LTs are considered to be prevented by
Katherine A Abrahams et al.
PloS one, 7(12), e52951-e52951 (2013-01-10)
Mycobacterium tuberculosis is a major human pathogen and the causative agent for the pulmonary disease, tuberculosis (TB). Current treatment programs to combat TB are under threat due to the emergence of multi-drug and extensively-drug resistant TB. Through the use of
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service![2-phenylimidazo[1,2-a]pyridine AldrichCPR](/deepweb/assets/sigmaaldrich/product/structures/281/247/6c2550a0-2f0c-4866-83d8-3c1fb039e165/640/6c2550a0-2f0c-4866-83d8-3c1fb039e165.png)
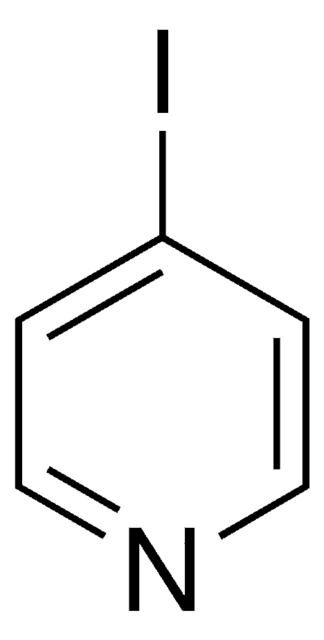
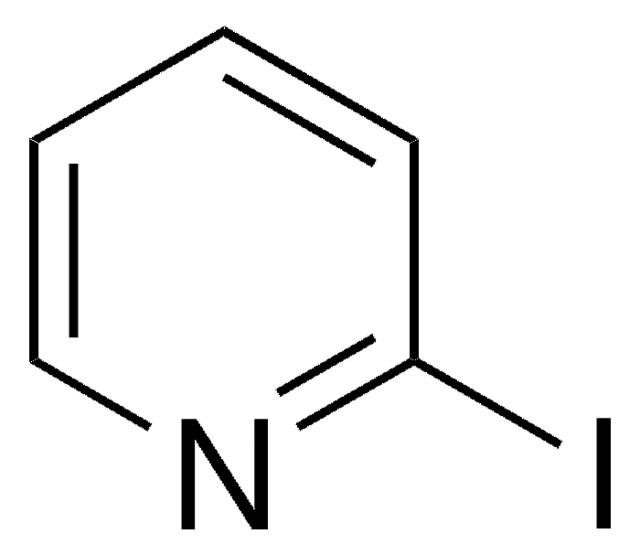
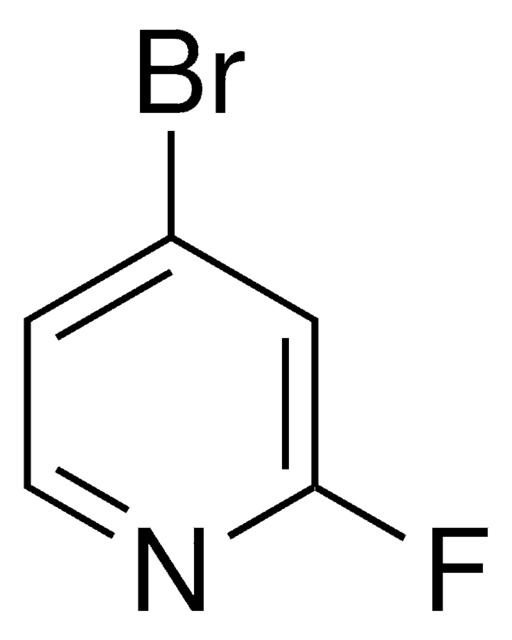
![Imidazo[1,2-a]pyrazine 97%](/deepweb/assets/sigmaaldrich/product/structures/370/804/1712d71f-52fb-4758-9a22-85b6c96cd4e8/640/1712d71f-52fb-4758-9a22-85b6c96cd4e8.png)
![Imidazo[1,2-a]pyrimidine AldrichCPR](/deepweb/assets/sigmaaldrich/product/structures/187/001/4862c14e-bec7-4475-85a5-f178e48ff60f/640/4862c14e-bec7-4475-85a5-f178e48ff60f.png)