90081
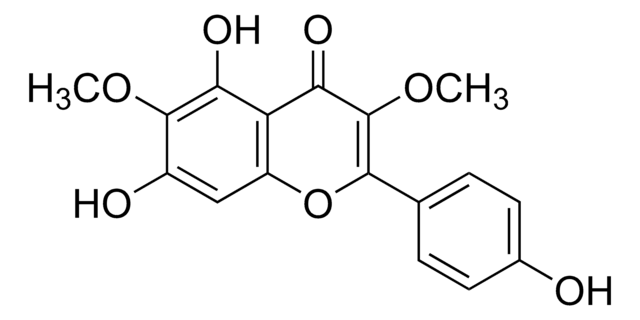
3-O-Methylquercetin
≥97% (HPLC)
Synonym(s):
5,7,3′ ,4′ -Tetrahydroxy-3-methoxyflavone, Quercetin 3-O-methyl ether
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C16H12O7
CAS Number:
Molecular Weight:
316.26
Beilstein:
324509
MDL number:
UNSPSC Code:
12352202
PubChem Substance ID:
NACRES:
NA.32
Recommended Products
Assay
≥97% (HPLC)
form
powder
SMILES string
COC1=C(Oc2cc(O)cc(O)c2C1=O)c3ccc(O)c(O)c3
InChI
1S/C16H12O7/c1-22-16-14(21)13-11(20)5-8(17)6-12(13)23-15(16)7-2-3-9(18)10(19)4-7/h2-6,17-20H,1H3
InChI key
WEPBGSIAWZTEJR-UHFFFAOYSA-N
Biochem/physiol Actions
3-O-Methylquercetin significantly inhibits cyclic adenosine monophosphate- (cAMP-) and cyclic guanosine monophosphate- (cGMP-) phosphodiesterase activity. It possess anti-inflammatory, bronchodilating properties and used in treatment of asthma. It suppresses the total inflammatory cells, tumor necrosis factor-α (TNF-α) and attenuates the production of interleukins.
3-O-Methylquercetin is a metabolite in flavone and flavonol biosynthesis. It is a naturally occurring compound present in various plants, and was shown to have potent anticancer-promoting, antioxidant, antiallergy, and antimicrobial activity, and showed strong anti-viral activity inhibition of tomato ringspot virus.
Packaging
Bottomless glass bottle. Contents are inside inserted fused cone.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Oral
Storage Class Code
6.1C - Combustible, acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Hyang Dok-Go et al.
Brain research, 965(1-2), 130-136 (2003-02-20)
The flavonoids quercetin, (+)-dihydroquercetin, and quercetin 3-methyl ether were isolated from the ethyl acetate fractions of the fruits and stems of Opuntia ficus-indica var. saboten. In the present study, we evaluated their protective effects against oxidative neuronal injuries induced in
L Van Puyvelde et al.
Journal of natural products, 52(3), 629-633 (1989-05-01)
3,5-Dihydroxy-6,7,8-trimethoxyflavone, 3-O-methylquercetin, and helichrysetin were isolated from the flowers of the Rwandese medicinal plant, Helichrysum odoratissimum. Because of inconsistencies of the mp of the latter chalcone, a synthesis of helichrysetin was developed. 3-O-Methylquercetin was shown to be an active principle
Mechanisms of suppression of nitric oxide production by 3-O-methylquercetin in RAW 264.7 cells
Jiang JS, et al.
Journal of Ethnopharmacology, 103(2), 281-287 (2006)
C Angeloni et al.
Biochimie, 89(1), 73-82 (2006-10-19)
The aim of this study was to investigate the potential of quercetin and two of its "in vivo" metabolites, 3'-O-methyl quercetin and 4'-O-methyl quercetin, to protect H9c2 cardiomyoblasts against H(2)O(2)-induced oxidative stress. As limited data are available regarding the potential
3-O-methylquercetin more selectively inhibits phosphodiesterase subtype 3
Ko WC, et al.
Planta Medica, 69(04), 310-315 (2003)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service