All Photos(1)
About This Item
Empirical Formula (Hill Notation):
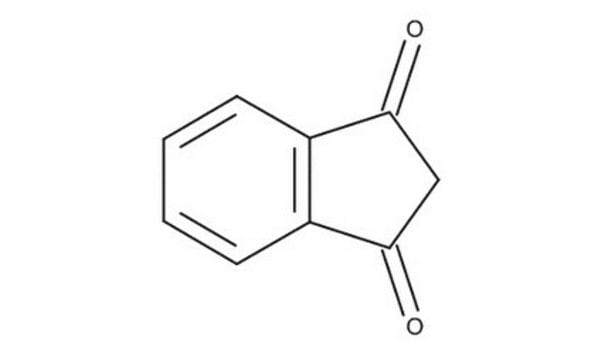
C9H6O2
CAS Number:
Molecular Weight:
146.14
Beilstein:
1210061
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
mp
129-132 °C (lit.)
SMILES string
O=C1CC(=O)c2ccccc12
InChI
1S/C9H6O2/c10-8-5-9(11)7-4-2-1-3-6(7)8/h1-4H,5H2
InChI key
UHKAJLSKXBADFT-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Abdolmajid Bayandori Moghaddam et al.
Chemical & pharmaceutical bulletin, 54(10), 1391-1396 (2006-10-04)
This is an environmentally friendly method in the field of electroorganic reactions under controlled potential electrolysis, without toxic reagents at a carbon electrode in an undivided cell which involves the (EC) mechanism reaction and comprises two steps alternatively; (i) electrochemical
A Schulman et al.
Journal of the American Veterinary Medical Association, 188(4), 402-405 (1986-02-15)
Five cases of coagulopathy caused by consumption of indanedione (diphacinone)-based rodenticides are reported. In each case, acute onset of lethargy and respiratory distress were the predominant initial clinical signs. Thoracic radiography revealed pulmonary edema, pleural effusion, and/or pericardial effusion as
A R Murthy et al.
Journal of medicinal chemistry, 28(11), 1591-1596 (1985-11-01)
A series of 2-substituted indan-1,3-dione derivatives, including alkyl (C-1-C-5), mono- and disubstituted phenyl, and other 2-aryl derivatives, were tested for hypolipidemic activity of CF1 male mice at 20 mg/kg per day. These derivatives reduced both serum cholesterol and triglycerides after
V Violet Dhayabaran et al.
Luminescence : the journal of biological and chemical luminescence, 32(7), 1339-1348 (2017-05-19)
A novel Schiff base, (S,Z)-4-(methylthio)-2-((3-oxo-2,3-dihydro-1H-inden-1-ylidene)amino)butanoic acid (L) and four M(II) complexes (where M = Co, Cu, Ni and Zn) were synthesized and characterized. The DNA-binding characteristics of the complexes were investigated using various spectroscopic methods and viscosity measurements. Analysis of the results
I H Hall et al.
Anticancer research, 14(5A), 2053-2058 (1994-09-01)
N-Substituted indan-1.3-diones have proven to be potent cytotoxic agents effective against the growth of single cell leukemia tumors and cell lines derived from solid tumors. A number of the derivatives were active against growth of solid tumors e.g. colon, lung
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service