B5386
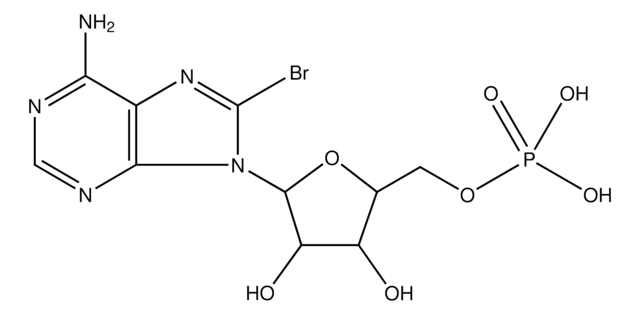
8-Bromoadenosine 3′,5′-cyclic monophosphate
≥97% (HPLC), powder, protein kinase A activator
Synonym(s):
8-Br-A-3:5-MP, 8-Br-cAMP, 8-Bromo-cAMP
About This Item
Recommended Products
product name
8-Bromoadenosine 3′,5′-cyclic monophosphate, ≥97% (HPLC)
Assay
≥97% (HPLC)
form
powder
mp
254 °C (dec.) (lit.)
solubility
aqueous base: soluble, clear
storage temp.
−20°C
SMILES string
Nc1ncnc2n([C@@H]3O[C@@H]4COP(O)(=O)O[C@H]4[C@H]3O)c(Br)nc12
InChI
1S/C10H11BrN5O6P/c11-10-15-4-7(12)13-2-14-8(4)16(10)9-5(17)6-3(21-9)1-20-23(18,19)22-6/h2-3,5-6,9,17H,1H2,(H,18,19)(H2,12,13,14)/t3-,5-,6-,9-/m1/s1
InChI key
DVKQVRZMKBDMDH-UUOKFMHZSA-N
Looking for similar products? Visit Product Comparison Guide
Application
- to treat H295R cells as positive controls for CYP19 induction
- as a membrane permeable cAMP analog to study its effect on short-circuit current (Isc)
- to investigate its potential as an inducer of differentiation in Wharton′s jelly-derived mesenchymal stem cells (WJ-MSCs)
Biochem/physiol Actions
Features and Benefits
Other Notes
Caution
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Cyclic nucleotides like cAMP modulate cell function via PKA activation and ion channels.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service