A0760
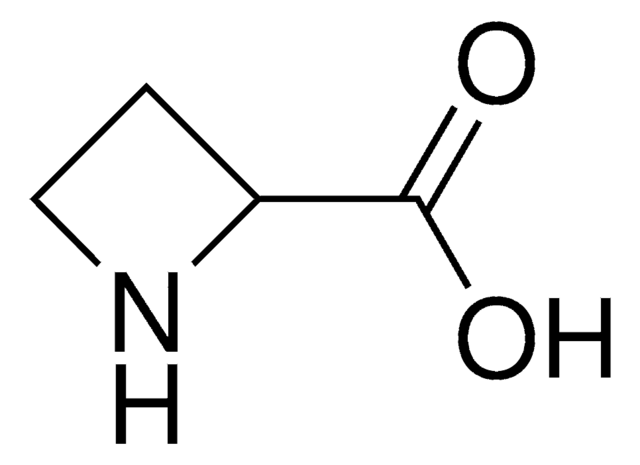
L-Azetidine-2-carboxylic acid
≥99%
Synonym(s):
(S)-Azetidine-2-carboxylic acid
Sign Into View Organizational & Contract Pricing
All Photos(4)
About This Item
Empirical Formula (Hill Notation):
C4H7NO2
CAS Number:
Molecular Weight:
101.10
Beilstein:
80680
EC Number:
MDL number:
UNSPSC Code:
12352209
PubChem Substance ID:
NACRES:
NA.32
Recommended Products
Quality Level
Assay
≥99%
form
powder
SMILES string
OC(=O)[C@@H]1CCN1
InChI
1S/C4H7NO2/c6-4(7)3-1-2-5-3/h3,5H,1-2H2,(H,6,7)/t3-/m0/s1
InChI key
IADUEWIQBXOCDZ-VKHMYHEASA-N
Looking for similar products? Visit Product Comparison Guide
General description
L-Azetidine-2-carboxylic acid is a non-protein amino acid and teratogenic agent. It is toxic in nature.
Application
L-Azetidine-2-carboxylic acid has been used as a:
- collagen synthesis inhibitor
- protein folding antagonist
- as a standard in liquid chromatography-mass spectrometry
Biochem/physiol Actions
Azetidine-2-carboxylic acid (AZC) triggers protein aggregation or upregulates the expression of an aggregation-prone mutant protein, upon interference with nascent protein folding.
L-Azetidine-2-carboxylic acid is an inhibitor of collagen synthesis that is anti-angiogenic.
L-Azetidine-2-carboxylic acid is an inhibitor of collagen synthesis that is anti-angiogenic. It is a four-membered ring analog of L-proline that causes protein misconstruction when incorporated instead of proline.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Lot/Batch Number
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Azetidine-2-carboxylic acid in garden beets (Beta vulgaris)
Rubenstein E, et al.
Phytochemistry, 67(9), 898-903 (2006)
Asmita Ghosh et al.
Cellular and molecular life sciences : CMLS, 76(8), 1605-1621 (2019-01-27)
The proteostasis network (PN) comprises a plethora of proteins that are dedicated to aid in protein folding and maintenance; some with overlapping functions. Despite this, there are multiple pathophysiological states associated with depletion of chaperones. This is counter-intuitive, assuming cells
Kate Samardzic et al.
Amino acids, 51(8), 1221-1232 (2019-07-16)
In addition to the 20 protein amino acids that are vital to human health, hundreds of naturally occurring amino acids, known as non-proteinogenic amino acids (NPAAs), exist and can enter the human food chain. Some NPAAs are toxic through their
Ascorbic acid promotes the stemness of corneal epithelial stem/progenitor cells and accelerates epithelial wound healing in the cornea
Chen J, et al.
Stem Cells Translational Medicine, 6(5), 1356-1365 (2017)
The anticancer drug AUY922 generates a proteomics fingerprint that is highly conserved among structurally diverse Hsp90 inhibitors
Voruganti S, et al.
Journal of Proteome Research, 12(8), 3697-3706 (2013)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service