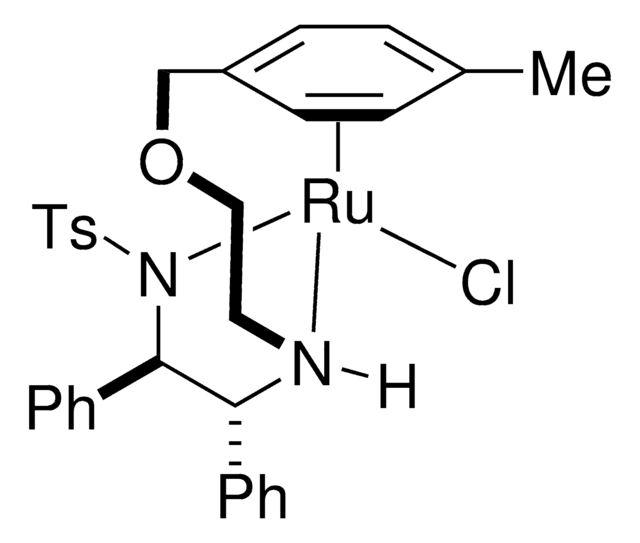
703907
RuCl(p-cymene)[(R,R)-Ts-DPEN]
Synonym(s):
[N-[(1R,2R)-2-(Amino-κN)-1,2-diphenylethyl]-4-methylbenzenesulfonamidato-κN]chloro[(1,2,3,4,5,6-η)-1-methyl-4-(1-methylethyl)benzene]-ruthenium
About This Item
Recommended Products
form
powder
optical activity
[α]20/D −116°, c = 0.1 in chloroform
mp
215 °C
functional group
amine
phenyl
storage temp.
2-8°C
SMILES string
CC(C)c1ccc(C)cc1.Cc2ccc(cc2)S(=O)(=O)N([Ru]Cl)[C@@H]([C@H](N)c3ccccc3)c4ccccc4
InChI
1S/C21H21N2O2S.C10H14.ClH.Ru/c1-16-12-14-19(15-13-16)26(24,25)23-21(18-10-6-3-7-11-18)20(22)17-8-4-2-5-9-17;1-8(2)10-6-4-9(3)5-7-10;;/h2-15,20-21H,22H2,1H3;4-8H,1-3H3;1H;/q-1;;;+2/p-1/t20-,21-;;;/m1.../s1
InChI key
AZFNGPAYDKGCRB-AGEKDOICSA-M
Looking for similar products? Visit Product Comparison Guide
General description
Application
- Quinolines
- N-Alkyl ketimines
- Antitumor and antiproliferative derivatives of natural products isolated from bacteria
- Hydroxy arylaldehydes via Rap-Stoermer-enantioselective transfer hydrogenation
Catalyst with improved performance modified via microenvironment engineering of nanocages
Legal Information
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Hydrogenation, Asymmetric Catalysis, Binap, SEGPHOS®, Aldol reaction, Alkenylation, Arylation, Mannich reaction, Fluorination, Michael addition, Hydrosilylation, Cycloaddition, Takasago
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service![RuCl(p-cymene)[(S,S)-Ts-DPEN]](/deepweb/assets/sigmaaldrich/product/structures/596/849/f8e3d2d8-a02e-430e-b0ed-c208cea3a6fb/640/f8e3d2d8-a02e-430e-b0ed-c208cea3a6fb.png)
 90%](/deepweb/assets/sigmaaldrich/product/structures/688/786/21c1946c-cdc5-40d4-bce5-5b82b1239504/640/21c1946c-cdc5-40d4-bce5-5b82b1239504.png)
![RuCl2[(R)−DM−BINAP][(R)−DAIPEN]](/deepweb/assets/sigmaaldrich/product/structures/355/628/d82e5e5d-23fd-468a-87ca-db4c45677898/640/d82e5e5d-23fd-468a-87ca-db4c45677898.png)
![(R)-RuCl[(p-cymene)(BINAP)]Cl](/deepweb/assets/sigmaaldrich/product/structures/244/078/7a0bdab6-11cc-4030-bbe9-4f687a6a925a/640/7a0bdab6-11cc-4030-bbe9-4f687a6a925a.png)

 95%](/deepweb/assets/sigmaaldrich/product/structures/151/609/eeb99dc1-9ef2-49d8-b255-6b5e2519fee1/640/eeb99dc1-9ef2-49d8-b255-6b5e2519fee1.png)
![[(R,R)-Teth-TsDpen RuCl]](/deepweb/assets/sigmaaldrich/product/structures/307/962/59412599-3794-437d-8a39-b88a036b7ca1/640/59412599-3794-437d-8a39-b88a036b7ca1.png)
 90%](/deepweb/assets/sigmaaldrich/product/structures/423/199/db3d73e8-0552-4abe-bdc3-3f9877bea3e6/640/db3d73e8-0552-4abe-bdc3-3f9877bea3e6.png)