BCR311
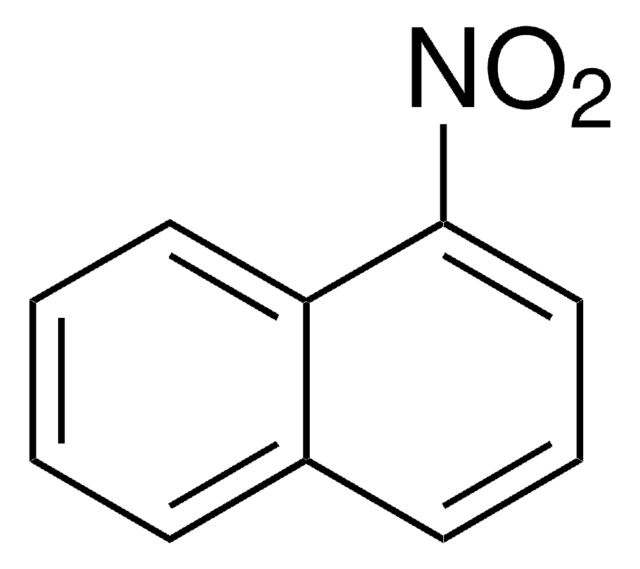
6-Nitrobenzo[a]pyrene
BCR®, certified reference material
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C20H11NO2
CAS Number:
Molecular Weight:
297.31
Beilstein:
2472924
MDL number:
UNSPSC Code:
41116107
PubChem Substance ID:
NACRES:
NA.24
Recommended Products
grade
certified reference material
Agency
BCR®
manufacturer/tradename
JRC
technique(s)
HPLC: suitable
gas chromatography (GC): suitable
format
neat
storage temp.
2-8°C
SMILES string
[O-][N+](=O)c1c2ccccc2c3ccc4cccc5ccc1c3c45
InChI
1S/C20H11NO2/c22-21(23)20-16-7-2-1-6-14(16)15-10-8-12-4-3-5-13-9-11-17(20)19(15)18(12)13/h1-11H
InChI key
NMMAFYSZGOFZCM-UHFFFAOYSA-N
General description
6-Nitrobenzo[a]pyrene belongs to the class of nitrated-polycyclic aromatic hydrocarbons, found to be persistent in the environment. It is produced from direct sources such as diesel, gasoline exhaust and by the gas-phase reactions of polycyclic aromatic hydrocarbons (PAHs) with oxides of nitrogen.
Analysis Note
For more information please see:
BCR311
BCR311
Legal Information
BCR is a registered trademark of European Commission
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Lot/Batch Number
Sorry, we don't have COAs for this product available online at this time.
If you need assistance, please contact Customer Support.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
M Sala et al.
Carcinogenesis, 8(4), 503-507 (1987-04-01)
Two nitroaromatics, 6-nitrobenzo[a]pyrene (6-N-BaP) and 6-nitrochrysene (6-N-CRY), and the corresponding parent hydrocarbons, benzo[a]pyrene (BaP) and chrysene (CRY), were studied in in vitro transformation assays with Syrian hamster embryo (SHE) cells, BALB/3T3 and C3H10T1/2 mouse cell lines. The three cell systems
Metabolism of 1-nitropyrene and 6-nitrobenzo(a)pyrene by intestinal microflora.
C E Cerniglia
Progress in clinical and biological research, 181, 133-137 (1985-01-01)
B S Hass et al.
Mutation research, 171(2-3), 123-129 (1986-08-01)
The mononitro-substituted isomers of benzo[a]pyrene (B[a]P), 1-, 3- and 6-nitrobenzo[a]pyrene (NB[a]P), are environmental pollutants and are metabolized to mutagens in Salmonella by rat-liver homogenate postmitochondrial supernatant (S9) fractions. In this study, activation of these compounds to mutagens was investigated using
G C Millner et al.
Journal of toxicology and environmental health, 19(4), 519-530 (1986-01-01)
The fungal metabolism of the potent mutagenic and carcinogenic nitropolycyclic aromatic hydrocarbon (nitro-PAH) 6-nitrobenzo[a]pyrene (6-NO2-BaP) was investigated. Cunninghamella elegans was incubated with 6-NO2-BaP for periods ranging between 1 and 7 d, and the metabolites formed were separated by high-performance liquid
Binary mixtures containing isomers of nitrobenzo[a]pyrene induce greater-than-additive mutational responses in Salmonella typhimurium. I. Analysis by the total concentration-proportional mixture model.
B S Hass et al.
Mutation research, 190(4), 247-252 (1987-04-01)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service