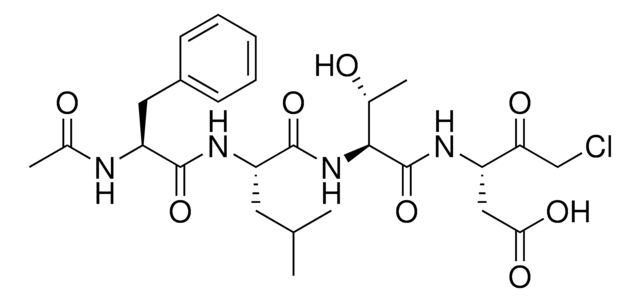
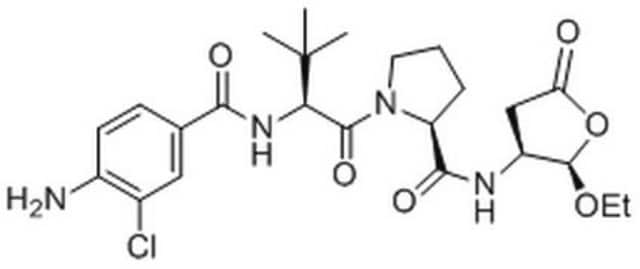
This product is a 5 mg/mL solution prepared in a 1:1 mixture of DMSO and Ethanol. The product is stored at 2-8°C and is released with a 4 years suggested retest schedule. The solution is quite stable refrigerated and freezing is not necessary. However, the pure powder product (N7143) is stable at -20°C for at least 3 months when dissolved in DMSO or Ethanol. Storage at less than -20°C has not been evaluated. Please see the link below for a sample Certificate of Analysis which includes the recommended retest date:
https://www.sigmaaldrich.com/certificates/sapfs/PROD/sap/certificate_pdfs/COFA/Q14/SML1779-1ML0000225533.pdf
Please see the link below to review the product datasheet for the powdered Nigericin, N7143:
https://www.sigmaaldrich.com/deepweb/assets/sigmaaldrich/product/documents/379/493/n7143pis.pdf